Archived Content
The National Institute of Mental Health archives materials that are over 4 years old and no longer being updated. The content on this page is provided for historical reference purposes only and may not reflect current knowledge or information.
Suspect Schizophrenia Genes Act Together to Thwart Working Memory
• Science Update
Two gene variants implicated in schizophrenia interact to degrade the brain's ability to process information, NIMH researchers have discovered. The interaction impaired working memory — retaining information from moment to moment. Such thinking problems are a hallmark of this severe mental illness that affects about one percent of the population.
Functional magnetic resonance imaging (fMRI) scans revealed an interaction between suspect versions of the genes that produced inefficient activity and poor connectivity in a key thinking circuit during a working memory task. Previous studies have shown that this circuit functions abnormally in schizophrenia.
The researchers scanned 29 healthy subjects while they performed working memory tasks — first easy and then hard — to gauge how the genes affected information processing in the circuit. The fMRI scanner produces pictures of how active parts of the brain are at any given moment by tracking the destination of oxygenated blood.
Joseph Callicott, M.D., Hao Yang Tan, M.D., and colleagues in the NIMH Genes, Cognition and Psychosis Program, reported on their findings in the July 24, 2007 Proceedings of the National Academy of Sciences.
"Much like a weak radio station drowned out by static, communications between brain cells were hampered by weakened signals relative to the background noise level in the circuit," explained Callicott.
The circuit handles communications between the executive hub at the front of the brain called the prefrontal cortex, and the middle areas important for processing information known as the parietal lobes.
While previous imaging studies had focused on individual genes, the new study shows that combinations of genes can thwart information processing, as likely occurs in schizophrenia. Schizophrenia is thought to stem from complex interactions among multiple genes and environmental factors. The COMT gene and GRM3 gene have been implicated in schizophrenia.
People typically inherit two copies of genes, one from each parent, so some people have two different versions while other have two of the same version. The two versions of the COMT gene, called "Val" and "Met," result from variation in just one letter of the genetic code. To maximize gene-influenced differences in brain activity, the researchers selected people who each had two copies of the "Val" gene version. These participants showed the working memory deficit if they also had two copies the GRM3 gene "A" version. Subjects with two copies of the COMT "Met" gene version were protected against the adverse interactive effects on working memory.
When the working memory task got harder, COMT "Val" carriers' brain activity soared in an area at the top of the prefrontal cortex; an area on the side of the cortex was also activated. But COMT "Met" carriers, who performed better on the task, activated only the top area, and to a lesser degree (see brain graphic below).
The over-activation in the top prefrontal area in the COMT "Val" carriers showed that it was processing information inefficiently — "much like a car with engine trouble that's working too hard and overheating," said Callicott. Activation of the side prefrontal area in the "Val" carriers appears to be the brain attempting to compensate for this, he added.
There was also evidence of poorer connections in the functioning of brain circuitry during working memory in those with the schizophrenia-related gene versions.
The "A" version of GRM3 is associated with weaker signals transmitted via the messenger chemical glutamate, while the COMT "Val" version is associated with weaker dopamine activity. The COMT "Met" version is marked by increased dopamine activity.
"Schizophrenia involves abnormalities in dopamine and glutamate," noted Callicott. "Interacting genes can affect the workings of key chemical messenger systems to impair the efficiency of communications signals in a circuit implicated in schizophrenia. Imaging these interactions deepens our understanding of disease mechanisms and may help point the way to improved treatment."
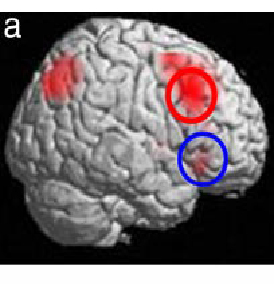
Functional MRI data superimposed on a 3-D anatomic MRI reconstruction of the brain shows areas (red) where activity differed during the easy and hard working memory tasks. Subjects with two copies of schizophrenia-related versions of two genes overactivated an area at the top (red circle) of the prefrontal cortex. They also activated an area at the side of the cortex (blue circle). By contrast, subjects with two copies of other common versions activated only the top (red circle) area - and to a lesser degree. The overactivation of the top area and likely compensatory activation of the side area indicates inefficient information processing, which was reflected in poorer performance on the task by those with the suspect gene versions.
Source: Joseph Callicott, M.D., NIMH Genes, Cognition and Psychosis Program
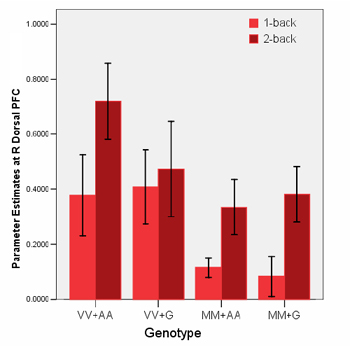
Subjects with two copies of the schizophrenia-implicated COMT gene "Val" version and the GRM3 gene "A" version (VV+AA) showed the highest activation in the top prefrontal cortex area (red circle in above graphic) during working memory tasks. This indicated the least efficient information processing, compared to other gene combinations. The darker red bars reflect heightened activation levels during the hard working memory task (2-back).
Source: Joseph Callicott, M.D., NIMH Genes, Cognition and Psychosis Program
Tan HY, Chen Q, Sust S, Buckholtz JW, Meyers JD, Egan MF, Mattay VS, Meyer-Lindenberg A, Weinberger DR, Callicott JH . Epistasis between catechol-O-methyltransferase and type II metabotropic glutamate receptor 3 genes on working memory brain function. Proc Natl Acad Sci U S A. 2007 Jul 24;104(30):12536-41. Epub 2007 Jul 16. PMID: 17636131
