Archived Content
The National Institute of Mental Health archives materials that are over 4 years old and no longer being updated. The content on this page is provided for historical reference purposes only and may not reflect current knowledge or information.
HIV-Infected Astrocytes Disrupt Blood-Brain Barrier, Contribute to Cognitive Impairment
Small Number of Infected Cells Have Profound Effects
• Science Update
Astrocytes, a type of support cell in the brain, that are infected with HIV show abnormal connections and functioning that disrupt the blood-brain barrier, according to an NIMH-funded study. This disruption may be responsible for the neurological disorders experienced by 40-60 percent of people infected with the virus, despite getting proper treatment. The study was published in the June 29, 2011, issue of the Journal of Neuroscience.
Background
Astrocytes are abundant throughout the central nervous system and may account for as much as half the brain’s mass. Among their many supportive functions, astrocytes help to maintain the blood-brain barrier through their “end feet,” which interact with structural components of the barrier. The blood-brain barrier separates the brain and spinal cord from circulating blood and restricts the passage of bacteria, viruses, and other potentially harmful substances into the central nervous system.
HIV primarily targets immune cells and infects astrocytes only in relatively small numbers, with no detectable increase in viral load; viral load is a measure of the severity of an infection and is sometimes used for diagnosis. The potential role of astrocytes in promoting HIV infection has not been well studied.
Eliseo Eugenin, Ph.D., of the Albert Einstein College of Medicine, and colleagues developed a model of the blood-brain barrier using HIV-infected astrocytes. They also compared the model to brain tissue samples taken at different time points post-infection from macaque monkeys infected with SIV (simian immunodeficiancy virus) , which is comparable to HIV in humans.
Results of the Study
In their model, the researchers observed that non-infected astrocytes sent projections through a thin filter (simulating the blood-brain barrier) and formed astrocyte end feet to connect with cells on the other side (see Figure 1). The resulting structure restricted the type and amount of substances that could pass through the filter and functioned comparably to the human blood-brain barrier.
In models containing a small number (around five percent) of infected astrocytes, the barrier structure was more permeable, and some cells on the other side of the filter died off. The researchers noted that infected astrocytes showed abnormal end foot connections, resulting in disruption of the barrier structure (see Figure 2). These irregularities could be reduced by blocking gap junction channels—pathways that link neighboring cells and allow for direct exchange of small molecules and thus may amplify harmful effects. Blocking certain signaling pathways, through which astrocyte end feet normally regulate the health and function of blood vessels, also reduced disruptions to the blood-brain barrier model.
In macaque brain tissue samples, infected astrocytes were surrounded by dead but uninfected cells. More severe SIV-related neurological symptoms were associated with more widespread cell death. The infected astrocytes themselves did not die.
Significance
The results reveal a potential mechanism through which small numbers of infected astrocytes may contribute to HIV-related neurological disorders without significantly increasing viral load. Namely, that infected astrocytes disrupt the structure and function of the blood-brain barrier by developing abnormal end foot connections and triggering cell death in uninfected, neighboring cells. This effect can be reduced by blocking gap junction channels or specific signaling pathways concentrated in astrocyte end feet.
Research on astrocytes and other viral reservoirs that may shield HIV from the beneficial effects of medications could inform efforts to improve on current treatments or develop methods to prevent HIV-related neurological disorders.
What’s Next
According to the researchers, future studies should explore the role of other cell types that are also involved in regulating the structure and function of the blood-brain barrier. Improved techniques that would allow specific components of gap junction channels to be targeted independently may also provide additional insight on how HIV-related damage spreads.
Reference
Eugenin EA, Clements JE, Zink MC, Berman JW. Human immunodeficiency virus infection of human astrocytes disrupts blood-brain barrier integrity by a gap junction-dependent mechanism . J Neurosci. 2011 Jun 29;31(26):9456-65. PubMed PMID: 21715610.
Figure 1: Illustration of Blood-Brain Barrier Model
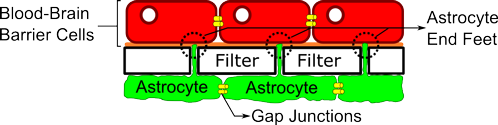
Source: Eliseo Eugenin, Ph.D., Albert Einstein College of Medicine
Figure 2: Enlarged Images of Astrocytes
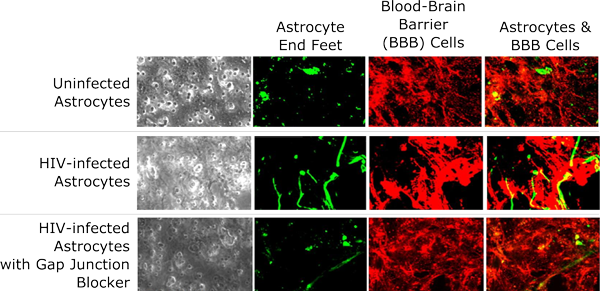
Figure 2: Enlarged microscope images of uninfected astrocytes, HIV-infected astrocytes, and HIV-infected astrocytes grown with gap junction blockers. Special staining techniques show how HIV infection results in irregular development of astrocyte end feet (green), disrupted growth of cells associated with the blood-brain barrier (red), and abnormal connections between the two. The bottom row demonstrates how gap junction blockers help to reduce these effects.
Source: Eliseo Eugenin, Ph.D., Albert Einstein College of Medicine
