Archived Content
The National Institute of Mental Health archives materials that are over 4 years old and no longer being updated. The content on this page is provided for historical reference purposes only and may not reflect current knowledge or information.
Exposure / Ritual Prevention Therapy Boosts Antidepressant Treatment of OCD
CBT Trumps Antipsychotic for Augmentation, Amending Current Guidelines
• Science Update
A form of behavioral therapy can augment antidepressant treatment of obsessive compulsive disorder (OCD) better than an antipsychotic, researchers have found. They recommend that this specific form of cognitive behavior therapy (CBT) – exposure and ritual prevention – be offered first to OCD patients who don’t respond adequately to treatment with an antidepressant alone, which is often the case. Current guidelines favor augmentation with antipsychotics.
In the controlled trial with 100 antidepressant-refractory OCD patients, 80 percent of those who received CBT augmentation responded, compared to 23 percent of those who received the antipsychotic risperidone, and 15 percent of those who received placebo pills. Forty-three percent experienced symptoms reduced to a minimal level following CBT treatment, compared to 13 percent for risperidone and 5 percent for placebo.
The study, published September 11, 2013 in JAMA Psychiatry, was led by NIMH grantees Helen Blair Simpson, M.D., Ph.D. , of Columbia University, in New York City; and Edna Foa, Ph.D. , of the University of Pennsylvania, Philadelphia.
In an accompanying editorial, grantees Kerry Ressler, M.D., Ph.D. and Barbara Rothbaum, Ph.D. , of Emory University, Atlanta, note that antidepressants are effective in treating only a subset of OCD patients. Similarly, antipsychotics appear to benefit only a subset of patients, such as those with tic-disorder related OCD.
While such medications transiently relieve symptoms of circuits gone awry, CBT is thought to act though different mechanisms that actually change the underlying workings of connections in those circuits, say the authors. For example, exposure to fear-provoking situations without experiencing the feared consequences appears to teach the brain’s habit-forming circuitry to unlearn dysfunctional behaviors, such as compulsive rituals. The only medication shown to enhance such emotional learning is D-cycloserine, which, unlike most other drug treatments, is given only just prior to therapy sessions.
Matthew Rudorfer, M.D., chief of the NIMH Somatic Treatments Program, which funded the study, said that in demonstrating how different patients respond best to different approaches, it helps to move the field toward the goal of more personalized treatment. In helping to explain how different treatment types work via different circuitry, the findings and analysis also dovetails with the objectives of the NIMH Research Domain Criteria (RDoC) project, according to Bruce Cuthbert, Ph.D., director of the NIMH Division of Adult Translational Research and Treatment Development.
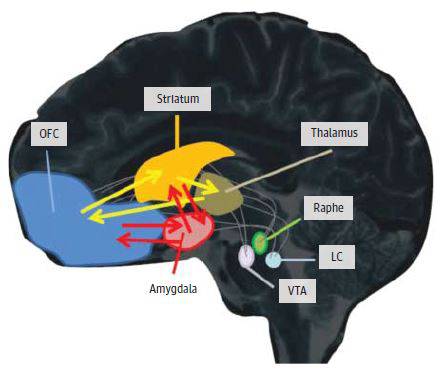
Source: Kerry Ressler, M.D., Ph.D., Barbara Rothbaum, Ph.D., Emory University
References
Cognitive-Behavioral Therapy vs Risperidone for Augmenting Serotonin Reuptake Inhibitors in Obsessive-Compulsive Disorder: A Randomized Clinical Trial . Simpson HB, Foa EB, Liebowitz MR, Huppert JD, Cahill S, Maher MJ, McLean CP, Bender J Jr, Marcus SM, Williams MT, Weaver J, Vermes D, Van Meter PE, Rodriguez CI, Powers M, Pinto A, Imms P, Hahn CG, Campeas R. JAMA Psychiatry. 2013 Sep 11. doi: 10.1001/jamapsychiatry.2013.1932. [Epub ahead of print] PMID:24026523
Augmenting Obsessive-Compulsive Disorder Treatment: From Brain to Mind . Ressler KJ, Rothbaum BO. JAMA Psychiatry. 2013 Sep 11. doi: 10.1001/jamapsychiatry.2013.2116. [Epub ahead of print] No abstract available. PMID:24026506
Clinical Trial: 00389493
