2013 Winter Inside NIMH

Welcome to the latest edition of Inside NIMH. This edition of the newsletter discusses recent funding opportunities and some new initiatives the Institute is considering for the future. We e-publish Inside NIMH after each meeting of the National Advisory Mental Health Council, which advises the Secretary of Health and Human Services; the Director, National Institutes of Health, and the Director of NIMH on all policies and activities relating to the conduct and support of mental health research, research training, and other programs of the Institute. In addition, check out the Director’s blog on our website for regular updates on timely topics at NIMH. I hope you find this information interesting and helpful. Please let us know if you have questions or comments on this edition.
Sincerely,
Thomas R. Insel, M.D.
Director, National Institute of Mental Health
If you wish to unsubscribe, subscribe, or change your email address, please contact the NIMH Webmaster or visit the Inside NIMH subscription page .
I. Message from the NIMH Director
Although we gaze forward with hope into 2013, we have all been affected by the tragic events that took place in December 2012 in Newtown, CT. NIMH was asked to contribute to the research and policy agenda for Vice President Biden’s Task Force to address issues of mental illness and gun violence, which informed President Obama’s January 16, 2013 Sandy Hook Response Plan . The Response Plan directs Secretary Sebelius of the Department of Health and Human Services (HHS), through the Centers for Disease Control and Prevention and other scientific agencies within HHS, to conduct or sponsor research into the causes of gun violence and the ways to prevent it. As the President noted in his remarks on January 16, people with serious mental illness (SMI), such as schizophrenia and bipolar disorder, compose only a small fraction of perpetrators of violent crimes, and are in fact more likely to be victims of violent crime themselves. Violence is associated with untreated psychosis, particularly when accompanied by symptoms of paranoia and substance abuse, but the risk of violence is reduced with appropriate treatment. The role of NIH in the Response Plan is evolving, and NIMH aims to continue supporting research on earlier diagnosis and quicker delivery of appropriate treatment not only to people with SMI, but to all individuals affected by mental illnesses.
When violence is associated with mental illness, it is much more often related to suicide rather than homicide. Approximately five percent of individuals with schizophrenia will die by suicide during their lifetime. In line with the HHS priority on suicide prevention, NIMH has increased its focus on the science of suicide. For example, the Army Study to Assess Risk and Resilience in Service-members (Army STARRS ) project is a Framingham-like study of military personnel. The goal of this five-year project is to identify, as rapidly as possible, risk and protective factors that will help the Army develop effective strategies to reduce rising suicide rates and to address associated mental health problems among soldiers. In fiscal year (FY) 2012, Army STARRS reached a number of milestones, including establishing survey sites at more than 70 locations around the world, surveying more than 100,000 Soldiers, and collecting more than 56,000 blood samples. This research will also help to increase our understanding of suicide in the overall population, leading to more effective prevention and treatment for service members and civilians alike.
Budget Overview
Review of FY 2012
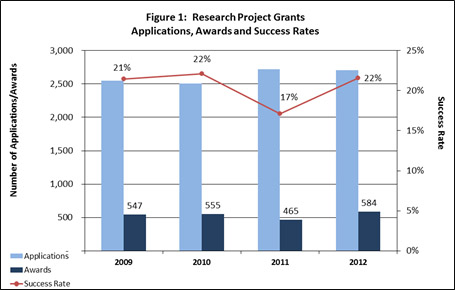
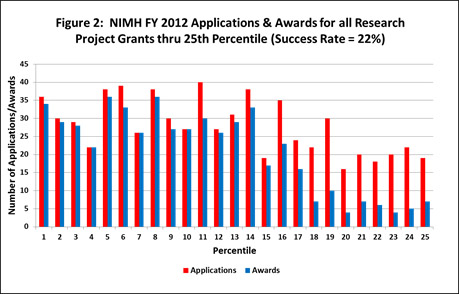
NIMH awarded 584 new and competing research project grants (RPGs) in FY 2012 and achieved an overall success rate of 22 percent (defined as number of RPG applications funded divided by the number of applications received; see Figures 1 and 2). This represents a significant increase over the 465 RPGs awarded in FY 2011, and is consistent with the FY 2008-2010 average of 558. NIMH awarded grants to 115 new principal investigators and achieved a success rate of 25 percent for early stage investigators (ESIs).
FY 2013 Budget
Although FY 2013 began on October 1, 2012, there is still considerable uncertainty about the NIH and NIMH budgets. In order to continue in the absence of a formal FY 2013 budget appropriation, President Obama signed a continuing resolution (CR) on September 27, 2012. The CR continues government operations through March 27, 2013 at the FY 2012 level, plus 0.6 percent.
While operating under a CR, we are issuing non-competing research grant awards at a level below that indicated on the most recent Notices of Award (generally up to 90 percent of the previously committed level). As in previous CRs, we look forward to upward adjustments after the final appropriation is enacted later in the fiscal year.
I have heard a lot of concern about the change in the NIH grant resubmission policy , allowing only one resubmission for each grant application. Sally Rockey, PhD, NIH’s Deputy Director for Extramural Research, recently reported the findings from an in-depth analysis of these changes in her “Rock Talk ” blog. Since the elimination of A2 applications, a higher proportion of A0 applications are being awarded compared to A1 applications, and the average time to award from submission of A0 applications has been reduced from 93 weeks to 56 weeks. Further, the time-to-award for new investigators is not appreciably longer than that of other applicants. This policy was developed as part of NIH efforts to enhance peer review, and it appears that this policy is meeting its goals to enable NIH to fund as much meritorious science as possible in as short a time period as possible.
New and Notable
A selection of the Institute’s most recently funded projects that exemplify our efforts to accelerate mental health research and to advance the NIMH Strategic Plan:
- Using a multidisciplinary and integrated translational approach, Peter Penzes, PhD , (Northwestern University at Chicago) is examining rare genetic mutations found in individuals with schizophrenia, and determining the effects of these mutations on neuronal plasticity. Data generated from this study may provide new insights into pathways that underlie abnormal brain connectivity in schizophrenia that will allow identification of therapeutic targets.
- Through an innovative integration of behavioral, biochemical, and brain imaging approaches, Diego Pizzagalli, PhD , (McLean Hospital) is examining early life stress and depression. Dr. Pizzagalli aims to provide a better understanding of neurobiological basis of outcomes related to childhood sexual abuse. The findings from this study could contribute to developing more targeted therapeutic interventions and identifying individuals at risk for psychopathology at a younger age.
- Martha Sajatovic, MD , (Case Western Reserve University) aims to improve outcomes for individuals with serious mental illness (SMI) who have diabetes, a major source of premature mortality in these individuals. Dr. Sajatovic will test the effectiveness of the Targeted Training in Illness Management (TTIM) intervention, which utilizes nurse and peer educators and is designed to overcome the complexities associated with co-management of SMI and medical disorders. If successful, TTIM could be easily adapted to related medical conditions such as metabolic syndrome and obesity, and implemented broadly in primary care settings.
- Kafui Dzirasa, MD, PhD , (Duke University), an early stage investigator, aims to characterize the neural circuits for complex behaviors, which may be useful to understand individual differences mediating susceptibility or resistance to the negative effects of chronic stress. Dr. Dzirasa will use an innovative combination of electrophysiological techniques to characterize the circuit-based mechanisms that mediate resistance to the negative effects of stress. This strategy may reveal new circuit-based targets for therapeutic intervention for mood and anxiety disorders.
For more information on these and other grants selected for funding, please visit the NIH RePORTER website .
Finally, I would like to welcome Susan Amara, PhD, as the new Scientific Director of the NIMH Division of Intramural Research Programs. Dr. Amara received her degree in Physiology and Pharmacology from the University of California, San Diego, and joined us from the University of Pittsburgh, School of Medicine. While serving as Scientific Director, Dr. Amara will continue her research in her new lab at NIMH, the Laboratory of Molecular and Cellular Neurobiology, in the Section on Molecular and Cellular Signaling.
Return to Top
II. New Announcements about Funding Opportunities
Each week, NIH electronically distributes the NIH GUIDE , a listing of all NIH Funding Opportunity Announcements (FOAs), which include requests for applications (RFAs), program announcements (PAs), and important notices for the scientific community. Below is a selection of recently issued FOAs in which NIMH participates. The Research and Funding page on the NIMH website has links to listings of all NIMH FOAs and other resources.
Note: You can subscribe to the NIMH Funding Opportunities LISTSERV to receive the latest information about RFAs and other research funding opportunities from NIMH, as well as administrative updates and changes to grant policies and procedures. You can also subscribe to a separate LISTSERV to receive weekly emails of the NIH GUIDE .
NIMH-Administered Requests for Applications
Leveraging Existing Natural Experiments to Advance the Health of People with Severe Mental Illness
Many state and local health authorities have implemented innovative services interventions to reduce several identified health risk factors and premature mortality in people with severe mental illness, although their effectiveness in this population, and the degree of adaptation needed for effectiveness, remain unknown. NIMH seeks to leverage these existing natural experiments by building research capacity for subsequent rigorous testing of services interventions already implemented at state and local levels for adults, youth or children. This FOA will support one-year R24 grants for research planning activities to develop the infrastructure needed to enable subsequent testing of existing innovative services interventions that aim to reduce the prevalence and magnitude of common modifiable health risk factors related to shortened lifespan in people with SMI. See the related Notice NOT-MH-13-010 .
- Release date: January 7, 2013; Expiration date: March 12, 2013
- R24 announcement (RFA-MH-13-140 )
Clinical Neuroscience and Entertainment Software Pilot Partnership Program to Develop Neuropsychiatric Interventions
This FOA utilizes the Small Business Innovation Research Program (SBIR) award mechanism to support the development of highly engaging cognitive training interventions delivered through computers and/or gaming platforms that are targeted to the treatment of neuropsychiatric disorders, autism and/or HIV Associated Neurocognitive Disorders (HAND), with the goal of improving real-world functioning of patients. This initiative specifically supports small businesses with development and commercial experience in the entertainment software industry to partner with clinical neuroscientists experienced with cognitive training.
- Release date: January 16, 2013; Expiration date: April 10, 2013
- R43/R44 announcement (RFA-MH-14-010 )
Biobehavioral Research Awards for Innovative New Scientists (BRAINS)
The BRAINS award is intended to support the research and research career development of outstanding scientists who are in the early, formative stages of their careers and who plan to make a long term career commitment to research in specific mission areas of NIMH. This award seeks to assist these individuals in launching an innovative clinical, translational, basic or services research program that holds the potential to profoundly transform the understanding, diagnosis, treatment, or prevention of mental disorders. Each year the BRAINS program will focus on a specific area of research and/or research career development need. For FY 2013 and FY 2014, the BRAINS program will focus on the research priorities and gap areas identified in the NIMH Strategic Plan and the Research Domain Criteria (RDoC) project.
- Release date: June 18, 2012; Expiration date: October 24, 2013
- R01 announcement (RFA-MH-13-110 )
NIMH-Collaborative Requests for Applications
Regional Comprehensive Metabolomics Resource Cores (RCMRC)
This FOA invites cooperative agreement applications to establish RCMRCs to increase the national capacity for utilizing metabolomics in biomedical research. To facilitate the biological and clinical application of metabolomics, technology service cores, pilot/feasibility projects, training activities, and outreach activities will be supported.
- Release date: December 2, 2011; Expiration date: February 16, 2013
- U24 announcement (RFA-RM-11-016 )
Exceptional Unconventional Research Enabling Knowledge Acceleration (EUREKA) for Neuroscience and Disorders of the Nervous System
This FOA solicits R01 applications addressing exceptionally novel hypotheses and/or remarkably difficult problems in neuroscience and disorders of the nervous system. This announcement is for support of new rather than ongoing projects, and is not intended for pilot research. The proposed research may have a high risk of failure, but it must promise results with especially high impact should it be successful. The research should be groundbreaking, innovative, original and/or unconventional, with the potential to solve important problems or open new areas for investigation.
- Release date: December 7, 2012; Expiration date: March 22, 2013
- R01 announcement (RFA-NS-13-007 )
NIH Revision Applications for Research Relevant to the Family Smoking Prevention and Tobacco Control Act
NIH and the Food and Drug Administration (FDA) have formed an interagency partnership to foster research relevant to tobacco regulations. This FOA invites revision applications from investigators and institutions/organizations with active NIH-supported P30 project awards to support an expansion of the scope of approved and funded scientific research programs involving smoking and tobacco-related products and/or their constituents. Investigators and institutions/organizations that are on a funded extension are eligible to apply for a revision. Revision applications for projects that have not originally focused on tobacco use are welcome provided that the applicant team has the requisite scientific expertise and the proposed revision addresses the research priorities related to FDA regulatory authority that are identified in the FOA.
- Release date: December 18, 2012; Expiration date: March 27, 2013
- P30 announcement (RFA-OD-12-007 )
NIH Blueprint for Neuroscience Initiatives
The Neuroscience Blueprint for Neuroscience is a framework to enhance cooperative activities among 16 NIH Institutes, Centers, and Offices that support research on the nervous system. The Blueprint aims to develop research tools, resources, and training and to make them available to the neuroscience community.
Human Connectome Project
The Human Connectome Project (HCP) is developing and using state-of-the-art neuroimaging technology to explore the connectivity of the healthy adult human brain. By systematically imaging hundreds of individuals, the HCP will provide insight into how brain connections underlie brain function. In addition to brain imaging, the HCP will involve collection of DNA samples, demographic information, and behavioral data from the study participants. Together, these data inform about how brain connectivity is influenced by genetics and the environment. HCP investigators have nearly completed phase one of the project (developing an informatics platform to allow the research community to access the data, and prototyping the phenotyping tests to be administered, etc.). Phase two (data collection) has started, and a small data release occurred in October 2012. The first full data release will occur in February 2013. The pilot data collected so far has resulted in several publications.
NIH Common Fund Initiatives
The NIH Common Fund encourages collaboration and supports a series of exceptionally high impact, trans-NIH programs. These programs are supported by the Common Fund, and managed by the NIH Office of the Director in partnership with the various NIH Institutes, Centers and Offices. The following projects currently have active funding opportunities and/or notices for NIMH applicants:
Health Economics
NIH launched the Health Economics Program to address the evolving need for health economic research. The program goals include fostering the collection of data that will be most useful for health policy-relevant analysis; examining the economic effects of changes in incentives for consumers, providers and insurers; exploring the ways in which structure and organization on the supply side of the medical market affect health care spending and clinical outcomes; and investigating the potential of preventive measures to improve health and mitigate cost growth. In November 2012, the Common Fund released two new Health Economics FOAs:
Diffusion of Medical Technology and Effects on Outcomes and Expenditures
- Release date: November 21, 2012; Expiration date: March 1, 2013
- U01 announcement (RFA-RM-12-023 )
Determinants and Consequences of Personalized Health Care and Prevention
- Release date: November 21, 2012; Expiration date: March 1, 2013
- U01 announcement (RFA-RM-12-024 )
Enhancing the Genotype-Tissue Expression (GTEx) Project
The GTEx project aims to provide a resource to the scientific community for the study of human gene expression and regulation and their relationship to genetic variation. In particular, the goal of this initiative is to understand how genetic variation may control gene expression across organs, tissues, and individuals. This project is collecting and analyzing multiple tissues from human donors who are also comprehensively genotyped, in order to assess genetic variation within their genomes. By analyzing global RNA expression within individual tissues and treating the expression levels of genes as quantitative traits, genetic variants that are highly correlated with changes in gene expression can be identified as expression quantitative trait loci (eQTLs). This FOA will support molecular analyses of stored biospecimens from the GTEx project. Applicants are encouraged to address compelling scientific questions with molecular phenotypes that add value to the resource and take full advantage of the unique features of GTEx.
- Release date: November 16, 2012; Expiration date: March 29, 2013
- R01 announcement (RFA-RM-12-009 )
Return to Top
III. Future Research Directions
Concept Clearances for Potential New Research Initiatives
This listing of potential future initiatives is meant to provide the earliest possible alert to the field of our research interests and of potential upcoming announcements to solicit that research. While NIMH plans to proceed with these initiatives, their publication and timing are not certain and depend on sufficient funding. The titles and brief descriptions are consistent with the information available at the time of concept clearance. The resultant FOAs may differ from the concepts in the final wording of their titles or other aspects. To send questions about a specific concept, follow the "Submit Comments" link at the bottom of the description.
- Integration and Analysis of Diverse HIV-Associated Data
- Methodologies and Formative Work for Combination Prevention Approaches
- Assessing the Role of HIV Stigma in HIV Prevention and Care
Related Information
- Recent NAMHC-approved concepts
- Recent public venue-approved concepts
- Past NAMHC meetings (contains links to agendas, minutes, and Director’s Reports)
Summary of NIMH-Sponsored Scientific Meetings
Research workshops and scientific meetings are some of the best forums in which to identify research gaps and to stimulate new areas of mental health research. Below is a brief description of meetings that NIMH sponsored recently. Questions about a specific meeting can be addressed by the program contact listed in the meeting description. For announcements, summaries, agendas, and participant lists from past NIMH-sponsored meetings, conferences, workshops, and lectures, please visit the Scientific Meetings page.
- Research to Improve Health and Longevity of People with Severe Mental Illness
Return to Top
IV. Update on Electronic Research Administration (eRA) Activities
For more information on all these updates, please see the NIH eRA News and Events page .
Electronic Grant Application Submission News
Changes for multi-project applications.
- NIH released a Guide Notice (NOT-OD-12-161 ) concerning plans to make changes to the application process for multi-project applications. These changes include: 1) transitioning multi-project applications from the paper PHS398 grant application to the Standard Form 424 Research and Related application form; and, 2) simultaneously transitioning to electronic submission of these multi-project applications. The Application Submission System and Interface for Submission Tracking (ASSIST) will accommodate electronic submission of NIH’s multi-project applications. ASSIST will allow users to develop and store their applications in a secure online environment. The system will also provide many of the features that have been requested by users over the years, such as error checking the application against both Grants.gov and many agency business rules before submission, providing the assembled application for viewing before submission, providing applicants with access control by component (subproject), and more. Please note the timeline for this transition.
- NIMH is participating in the pilot testing of the new ASSIST electronic submission process with the renewal publications of the National Cooperative Drug Discovery/Development Groups (NCDDG) for the Treatment of Mental Disorders, Drug or Alcohol Addiction (UM1 and U19). The purpose of the NCDDG Program is to create multidisciplinary research groups or partnerships for the discovery of pharmacological agents to treat and study mental illness, drug or alcohol addiction. The objectives of this program are to: accelerate innovative drug discovery; develop pharmacologic tools for basic and clinical research on mental disorders, or drug or alcohol addiction; develop and validate models for evaluating novel therapeutics for mental disorders; and support early phase human clinical testing to rapidly assess the safety and efficacy of promising drug candidates and new indications for IND-ready agents for the treatment of mental disorders or alcohol addiction. NIMH, the National Institute on Drug Abuse (NIDA), and the National Institute on Alcohol Abuse and Alcoholism (NIAAA) encourage applications to advance the discovery, preclinical development, and proof of concept testing of new, rationally based candidate agents to treat mental disorders or drug or alcohol addiction, and to develop novel ligands as tools to further characterize existing or to validate new drug targets. Partnerships between academia and industry are strongly encouraged.
- Release date: January 11, 2013; First Application due date: February 22, 2013 (with subsequent due dates in the following June, October, and February)
- Expiration date: October 23, 2015
- UM1 announcement (PAR-13-087 )
- U19 announcement (PAR-13-086 )
Questions? Find Help at: http://grants.nih.gov/grants/ElectronicReceipt/support.htm . This help desk contact is the only way to document problems and become eligible for any special consideration by the Division of Receipt and Referral should you run into a system problem with Grants.gov or with eRA that is beyond your control.
Return to Top
V. Research Training and Career Development
Providing Research Experiences for Physicians and Medical Students from Diverse Backgrounds (PREP)
NIMH announces an administrative supplement program to provide funds for NIMH-supported research projects to support advanced research experiences for outstanding early career physicians and medical students from diverse backgrounds. The purpose of this program is to improve the diversity of the mental health research workforce by supporting and recruiting early-stage investigators from groups that have been shown to be underrepresented in science. For the purposes of this initiative, eligible candidates include individuals from underrepresented racial and ethnic groups and individuals with disabilities. NIMH expects that participation in this administrative supplement program will enhance the professional development of the participating individuals, sustain their career trajectory through research independence, and lead to scientific advances that will help transform the understanding and treatment of mental illness. Please see submission instructions which are intended to be used in conjunction with the Notice (NOT-MH-12-019 ) for this administrative supplement program.
NIMH Updates Policy for Mentored K Applications and Awards
In late October 2012, NIMH announced some changes in NIMH policy for mentored K applications and awards (K01, K08, and K23; NOT-MH-13-001 ). The policy modifications were made after a careful examination of trends in application inquiries and applications. As noted on the Research Training and Career Development web page, NIMH uses the K01, K08, and K23 programs to provide opportunities for early-stage investigators who need additional mentored research experience to develop their independent research careers. NIMH encourages individuals to complete mentored training, transition to research independence, and secure independent R01 funding before the period of eligibility as an early-stage investigator ends (i.e., 10 years after completion of the terminal degree or clinical training as defined by the NIH). Effective with new applications due on February 12, 2013 and thereafter, eligibility for NIMH K01, K08, and K23 mentored career development awards is limited to individuals with no more than six years of postdoctoral experience at the time of application (either the initial or resubmission application). NIMH will generally not consider applications requesting more than four years of K01, K08, or K23 support. Waivers to both limits will be considered on a case-by-case basis, with strong justification. Please direct any questions to an NIMH Training Officer.
We’re interested in feedback from the community; comments or suggestions related to NIMH’s support for research training and career development may be directed to NIMH_Training@mail.nih.gov.
Return to Top
VI. Recent NIMH Science News
The latest news and updates from NIMH-supported research:
- 50 years ago, President John F. Kennedy Sent a Special Message to Congress About the State of Mental Health (January 29, 2013)
- Leading Neurobiologist Appointed NIMH Scientific Director (January 24, 2013)
- Study Documents that Some Children Lose Autism Diagnosis (January 15, 2013)
- Guide Offers a Blueprint for End-of-Life Conversation With Youth (January 07, 2013)
- Emergency Department Suicide Screening Tool Accurately Predicts At Risk Youth (January 04, 2013)
- Different Genes, Same Risk Pathway in Schizophrenia (January 02, 2013)
- Stress-Resilience/Susceptibility Traced to Neurons in Reward Circuit (December 12, 2012)
- Experimental Agent Briefly Eases Depression Rapidly in Test (December 11, 2012)
- Psychotropic Medications Are Prescribed Appropriately Among U.S. Teens, National Study Finds (December 03, 2012)
- Switching Off a Specific Brain Region Can Alter Ingrained Habits in Rats (November 27, 2012)
- In-sync Brain Waves Hold Memory of Objects Just Seen (November 01, 2012)
- NIH Common Fund Announces Awards for Single Cell Analysis (October 15, 2012)
- Gene Variants Implicated in Extreme Weight Gain Associated with Antipsychotics (October 12, 2012)
- Many Teens Considering Suicide Do Not Receive Specialized Mental Health Care (October 12, 2012)
- Former NIMH Grantee Wins Nobel Prize for Chemistry (October 10, 2012)
Publicizing NIMH research is a communal responsibility — we need your help! Please help us spread the word about the results of NIMH funding by acknowledging our support of your research, for example, in journal articles (citing your NIMH award by number when possible) and other communications. NIMH has two primary methods of getting the word out: press releases and science updates. All releases and updates are posted to the Science News section of the NIMH Website. These are all also distributed to the public through the NIMH ListServ, which now has more than 20,000 subscribers
If you have a manuscript accepted for publication that describes an especially significant finding, please contact your NIMH Program Official to discuss the possibility of a news release or other forms of dissemination.
Return to Top
VII. Connect with NIMH
Our newest effort to reach our stakeholders is a service that allows you to subscribe for updates sent directly to your email inbox on the NIMH topics of your choice. In addition to our email newsletters and RSS updates, NIMH offers and about mental health topics, and has its own YouTube channel . We have also entered the world of Twitter , where we highlight Science Updates, Press Releases, and other timely matters. You can even find us on Facebook ! Be sure to read our Director’s Blog for insights into the latest topics in mental health research.
Check us out!
