Science News
- Novel Treatment Helps Children With Severe Irritability
-
A new study by NIMH researchers demonstrated the effectiveness, feasibility, and safety of exposure-based cognitive behavioral therapy for severe irritability and temper outbursts in children. The positive results set the foundation to continue exploring exposure therapy as a potential treatment for childhood irritability.
- Revolutionizing the Study of Mental Disorders
-
• 75th Anniversary
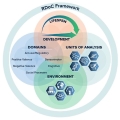
The Research Domain Criteria Initiative (RDoC) represented a new way to conceptualize the study of mental illnesses. In celebration of NIMH's 75th Anniversary, we reflect on the beginning and progress of this initiative.
- Study Reveals Potential Neural Marker for Social Impairment in Psychotic Disorders
-
Research funded by NIMH found a link between a low level of social interest among people with psychotic disorders and brain regions in the social motivation system.
- NIH Researchers Identify Brain Connections Associated With ADHD in Youth
-
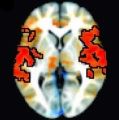
Researchers at the National Institutes of Health (NIH) have discovered that symptoms of attention-deficit/hyperactivity disorder (ADHD) are tied to atypical interactions between the brain’s frontal cortex and information processing centers deep in the brain.
- Life with Schizoaffective Disorder
-
• 75th Anniversary
In this lived experience feature story, Ray Lay describes the challenges of living with schizoaffective disorder and his later success in life.
- Dr. Joshua Gordon to Step Down as Director of the National Institute of Mental Health
-
On February 29, 2024, Joshua A. Gordon, M.D., Ph.D., announced his decision to end his tenure as the director of the National Institute of Mental Health.
- Intervention Reduces Likelihood of Developing Postpartum Anxiety and Depression by More Than 70%
-
Results from a large clinical trial funded by the National Institutes of Health show that an intervention for anxiety provided to pregnant women living in Pakistan significantly reduced the likelihood of the women developing moderate-to-severe anxiety, depression, or both six weeks after birth.
- Decades of Dedication and Collaboration: Unraveling the HIV Mystery
-
• 75th Anniversary
In celebration of NIMHs 75th anniversary, we reflect on decades of work by the institute to understand and eradicate HIV.
- Cognitive Behavioral Therapy Alters Brain Activity in Children With Anxiety
-
Researchers at the National Institute of Mental Health found that unmedicated children with anxiety disorders show widespread overactivation in brain functioning and that treatment with cognitive behavioral therapy led to a clinically significant drop in anxiety symptoms and improved brain functioning.
- Researchers Expand Understanding of Genetic Mechanisms Underlying Fragile X Syndrome
-
An NIMH-supported study of the 3D genome revealed widespread silencing of genes with important roles in brain function in fragile X syndrome and related disorders.