Archived Content
The National Institute of Mental Health archives materials that are over 4 years old and no longer being updated. The content on this page is provided for historical reference purposes only and may not reflect current knowledge or information.
Stress Hormone Receptors Less Adaptive in Female Brain
Findings in Rats Could Explain Women’s Increased Vulnerability to Disorders
• Science Update
A study in rats has revealed striking gender differences in the brain's stress response that could shed light on women's proneness to mood and anxiety disorders. Female rat brain cells were more sensitive to a key stress hormone than males', which could adapt to the hormone in a way female cells couldn't.
In the male brain under stress, many of the hormone's receptors retreated into the cell, making the brain less stress reactive. A molecular dance unique to the male brain, between the receptor and an enabling protein, accounted for its resilient adaptation. By contrast, in the female brain under stress, receptors remained exposed on neuron membranes and the brain stayed sensitive to the hormone.
"Although more research is necessary to determine whether this translates to humans, these findings may help to explain why women are twice as vulnerable as men to many stress-related disorders," explained NIMH grantee Rita Valentino Ph.D., of The Children's Hospital of Philadelphia.
A team of researchers led by Valentino and Debra Bangasser, Ph.D., reported on their discovery online June 15, 2010 in the journal Molecular Psychiatry.
Background
An understanding of why women experience more stress-related mental disorders like depression and PTSD has until now eluded science.
Corticotropin releasing factor (CRF), which acts as both a hormone and a neurotransmitter, is likely a key player. In response to a stressor, CRF binds to receptors on cells in an alarm center deep in the brainstem, called the locus ceruleus. This telegraphs heightened emotional arousal throughout the brain via the chemical messenger norepinephrine. Such hyper-arousal can be adaptive for brief periods, but not if it becomes chronic. Runaway CRF is a core feature of depression.
Previous studies suggested that this alarm system is more sensitive to CRF and stress in the female brain. To pinpoint how this works at the level of cells and molecules, Valentino, Bangasser and colleagues used antibodies and an electron microscope to see how the CRF receptor responds in male versus female rats — both unstressed and after exposure to a stressful swim.
Results
Even in the absence of any stress, the researchers found the female stress signaling system to be more sensitive from the start. CRF receptors had stronger connections, or coupling, with relay proteins inside the cell than those of male rats. So it took lower levels of CRF to activate neurons in the unstressed females compared to males. CRF levels that had no effect in males turned on cells in female rats.
After stress, CRF receptors remained exposed on the neuronal membrane in the female rat, allowing CRF to persist in its effect. In the stressed male rat, the receptors interacted with internal proteins called arrestins, enabling some to retreat into the cell's interior, where they couldn't bind with the hormone. Such receptor internalization helped the male brain adapt its sensitivity to the stressor. Although the arrestin proteins are present in the female neurons, the receptors did not interact with them.
Significance
In females, certain brain cells are more sensitive to CRF and less able to adapt to too much CRF. The greater coupling of CRF receptors to relay proteins and their inability to internalize could translate into a lower threshold for stress-induced activation of the alarm system. This could increase risk for chronic activation and impair the brain's ability to cope with high levels of CRF, as occurs in depression and PTSD, say the researchers. The study is the first to uncover sex differences at the level of receptor signaling, according to Valentino.
What's Next?
The next step is to examine the male and female CRF receptors for structural differences that might account for the functional differences, said Valentino. Since most rodent models of mood and anxiety disorders use males exclusively, the new findings of gender differences in stress signaling mechanisms call for a more gender-balanced approach — especially for disorders that disproportionately affect females. The gender differences in stress signaling should also be factored-in as medication treatments based on blocking the CRF receptor are developed, say the researchers.
Molecular Dance of CRF Receptors
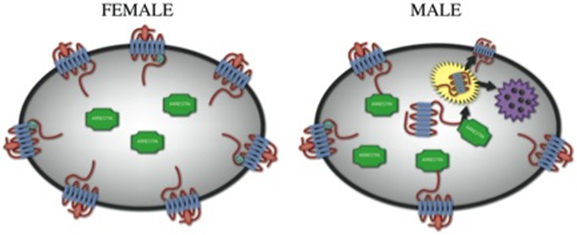
When the going gets tough inside a locus ceruleus neuron, it's the female brain that acts "macho." In response to a stressor, receptors for the stress hormone CRF remained exposed on the neuronal membrane in the female rat — taking the full hit. This increased CRF binding heightened the brain's stress reactivity. By contrast, in the stressed male rat, CRF receptors danced with internal proteins called arrestins (green), which enabled some to retreat into the cell's interior, where they couldn't bind with CRF. This adaptation — unique to the male brain — toned-down the neuron's stress sensitivity. Lack of such receptor internalization in the female brain could translate into impaired ability to cope with high levels of CRF — as occurs in depression and PTSD.
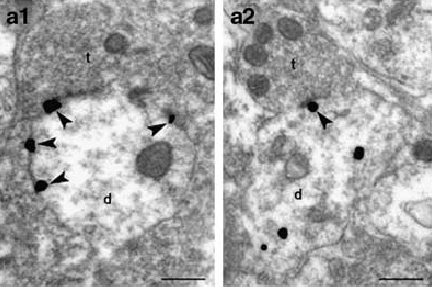
Electron microscope photo of unstressed male rat brain locus ceruleus (left) shows CRF receptors (black spots with arrows) on the cell membrane. Following exposure to a stressor (right), receptors have retreated into the interior of the cell (black spots without arrows). Such receptor internalization enables the male brain to regulate its sensitivity to stressors. This adaptation does not occur in the female brain, which could account for increased vulnerability to stress-related disorders.
Reference
Sex differences in corticotropin-releasing factor receptor signaling and trafficking: potential role in female vulnerability to stress-related psychopathology . Bangasser DA, Curtis A, Reyes BA, Bethea TT, Parastatidis I, Ischiropoulos H, Van Bockstaele EJ, Valentino RJ. Mol Psychiatry. 2010 Jun 15. [Epub ahead of print]PMID: 20548297
