Archived Content
The National Institute of Mental Health archives materials that are over 4 years old and no longer being updated. The content on this page is provided for historical reference purposes only and may not reflect current knowledge or information.
Our Brains Are Made of the Same Stuff, Despite DNA Differences
Gene expression databases reveal "consistent molecular architecture"
• Press Release
Despite vast differences in the genetic code across individuals and ethnicities, the human brain shows a “consistent molecular architecture,” say researchers supported by the National Institutes of Health. The finding is from a pair of studies that have created databases revealing when and where genes turn on and off in multiple brain regions through development.
“Our study shows how 650,000 common genetic variations that make each of us a unique person may influence the ebb and flow of 24,000 genes in the most distinctly human part of our brain as we grow and age,” explained Joel Kleinman, M.D., Ph.D., of the National Institute of Mental Health (NIMH) Clinical Brain Disorders Branch.
Kleinman and NIMH grantee Nenad Sestan, M.D., Ph.D. of Yale University, New Haven, Conn., led the sister studies in the Oct. 27, 2011 issue of the journal Nature.
“Having at our fingertips detailed information about when and where specific gene products are expressed in the brain brings new hope for understanding how this process can go awry in schizophrenia, autism and other brain disorders,” said NIMH Director Thomas R. Insel, M.D.
Both studies measured messenger RNAs or transcripts. These intermediate products carry the message from DNA, the genetic blueprint, to create proteins and differentiated brain tissue. Each gene can make several transcripts, which are expressed in patterns influenced by a subset of the approximately 1.5 million DNA variations unique to each of us. This unique set of transcripts is called our transcriptome – a molecular signature that is unique to every individual. The transcriptome is a measure of the diverse functional potential that exists in the brain.
Both studies found that rapid gene expression during fetal development abruptly switches to much slower rates after birth that gradually decline and eventually level off in middle age. These rates surge again as the brain ages in the last decades, mirroring rates seen in childhood and adolescence, according to one of the studies. The databases hold secrets to how the brain’s ever-changing messenger chemical systems, cells and development processes are related to gene expression patterns through development.
For example, if a particular version of a gene is implicated in a disorder, the new resources might reveal how that variation affects the gene’s expression over time and by brain region. By identifying even distant genes that may be turning on and off in-sync, the databases may help researchers discover whole modules of genes involved in the illness. They can also reveal how variation in one gene influences another’s expression.
Prefrontal cortex
Kleinman’s team focused on how genetic variations are linked to the expression of transcripts in the brain’s prefrontal cortex, the area that controls insight, planning and judgment, across the lifespan. They studied 269 postmortem, healthy human brains, ranging in age from two weeks after conception to 80 years old, using 49,000 genetic probes. The database on prefrontal cortex gene expression alone totals more than 1 trillion pieces of information, according to Kleinman.
Among key findings in the prefrontal cortex:
- Individual genetic variations are profoundly linked to expression patterns. The most similarity across individuals is detected early in development and again as we approach the end of life.
- Different types of related genes are expressed during prenatal development, infancy, and childhood, so that each of these stages shows a relatively distinct transcriptional identity. Three-fourths of genes reverse their direction of expression after birth, with most switching from on to off.
- Expression of genes involved in cell division declines prenatally and in infancy, while expression of genes important for making synapses, or connections between brain cells, increases. In contrast, genes required for neuronal projections decline after birth – likely as unused connections are pruned.
- By the time we reach our 50s, overall gene expression begins to increase, mirroring the sharp reversal of fetal expression changes that occur in infancy.
- Genetic variation in the genome as a whole showed no effect on variation in the transcriptome as a whole, despite how genetically distant individuals might be. Hence, human cortexes have a consistent molecular architecture, despite our diversity.
In previous studies, Kleinman and colleagues have found that all genetic variations implicated to date in schizophrenia are associated with transcripts that are preferentially expressed in the fetal brain. This adds to evidence that the disorder originates in prenatal development. By contrast, he and his colleagues are examining evidence that genetic variation implicated in affective disorders may be associated with transcripts expressed later in life. They are also extending their database to include all transcripts of all the genes in the human genome, examining 1000 post-mortem brains, including many of people who had schizophrenia or other brain disorders.
Multiple brain regions
Sestan and colleagues characterized gene expression in 16 brain regions, including 11 areas of the neocortex, from both hemispheres of 57 human brains that spanned from 40 days post-conception to 82 years – analyzing the transcriptomes of 1,340 samples. Using 1.4 million probes, the researchers measured the expression of exons , which combine to form a gene’s protein product. This allowed them to pinpoint changes in these combinations that make up a protein, as well as to chart the gene’s overall expression.
Among key findings:
- Over 90 percent of the genes expressed in the brain are differentially regulated across brain regions and/or over developmental time periods. There are also widespread differences across region and time periods in the combination of a gene’s exons that are expressed.
- Timing and location are far more influential in regulating gene expression than gender, ethnicity or individual variation.
- Among 29 modules of co-expressed genes identified, each had distinct expression patterns and represented different biological processes. Genetic variation in some of the most well-connected genes in these modules, called hub genes, has previously been linked to mental disorders, including schizophrenia and depression.
- Telltale similarities in expression profiles with genes previously implicated in schizophrenia and autism are providing leads to discovery of other genes potentially involved in those disorders.
- Sex differences in the risk for certain mental disorders may be traceable to transcriptional mechanisms. More than three-fourths of 159 genes expressed differentially between the sexes were male-biased, most prenatally. Some genes found to have such sex-biased expression had previously been associated with disorders that affect males more than females, such as schizophrenia, Williams syndrome, and autism.
The Sestan study was also funded by NIH’s National Institute on Child Health and Human Development, National Institute on Neurological Disorders and Stroke, and National Institute on Drug Abuse. Data for the Sestan study are posted at www.humanbraintranscriptome.org and at http://www.developinghumanbrain.org , as part of a larger ongoing study, BrainSpan, funded by NIMH under the American Recovery and Reinvestment Act to create an Atlas of Human Brain Development.
The Kleinman study data on genetic variability are accessible to qualified researchers at http://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/study.cgi?study_id=phs000417.v1.p1 , while the gene expression data can be found at http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc5GSE30272 . In addition, BrainCloud, a web browser application developed by NIMH to interrogate the Kleinman study data, can be downloaded at http://braincloud.jhmi.edu/.
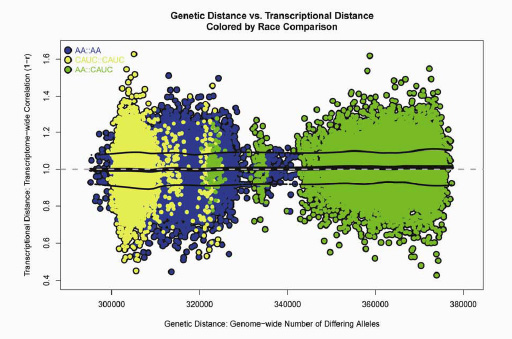
Our brains are all made of the same stuff. Despite individual and ethnic genetic diversity, our prefrontal cortex shows a consistent molecular architecture. For example, overall differences in the genetic code (“genetic distance”) between African -Americans (AA) and caucasians (cauc) showed no effect on their overall difference in expressed transcripts (“transcriptional distance”). The vertical span of color-coded areas is about the same, indicating that our brains all share the same tissue at a molecular level, despite distinct DNA differences on the horizontal axis. Each dot represents a comparison between two individuals. The AA::AA comparisons (blue) generally show more genetic diversity than cauc::cauc comparisons (yellow), because caucasians are descended from a relatively small subset of ancestors who migrated from Africa, while African Americans are descended from a more diverse gene pool among the much larger population that remained in Africa. AA::cauc comparisons (green) differed most across their genomes as a whole, but this had no effect on their transcriptomes as a whole.
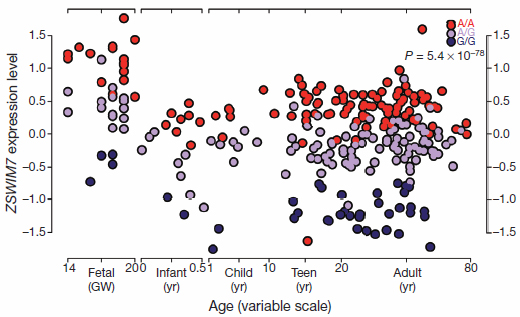
Individual genetic variation likely affects the expression of genes. For example, across all ages and ethnicities studied, expression of a gene called ZSWIM7 stays low, medium or high in the prefrontal cortex, depending on which of three versions (A/A, A/G, G/G) is inherited. The versions are created by a tiny variation in the letters of the genetic code (DNA) at a location in the gene called rs1045599.
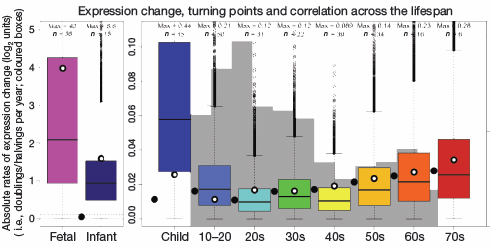
Overall gene expression plummets 5-fold in infancy and 90-fold in childhood from its prenatal peak. The decline levels-off during the middle years, but expression surges again in the last decades of life, as the brain ages. Note: The fetal/infant graph at left is based on a different scale than the lifespan graph at right, so the two are not visually comparable.
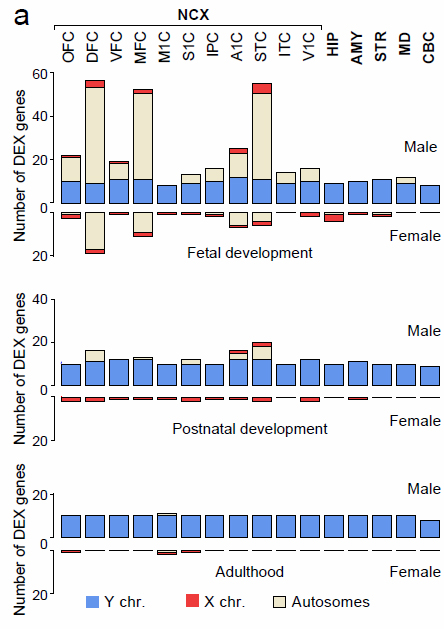
Males show more sex-biased gene expression. More genes differentially expressed (DEX) between the sexes were found in males than females, especially prenatally. Some genes found to have such sex-biased expression had previously been associated with disorders that affect males more than females, such as schizophrenia, Williams syndrome, and autism. Eleven of the brain areas shown are in the neocortex (NCX), or outer mantle.
Source: Nenad Sestan, M.D., Ph.D., Yale University Department of Neurobiology and Kavli Institute for Neuroscience
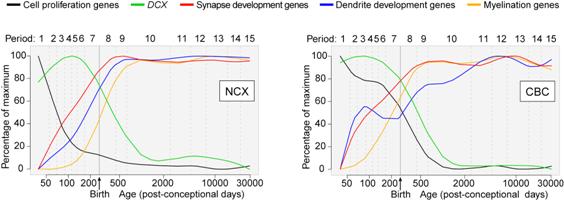
Profiling developmental processes. The expression data can be used to create trajectories for the expression of genes involved in particular processes, such as the development of synapses (structures that underlie communication between neurons). These expression trajectories can be compared for different regions, such as the NCX and cerebellar cortex (CBC).
Source: Nenad Sestan, M.D., Ph.D., Yale University Department of Neurobiology and Kavli Institute for Neuroscience
References
Colantuoni c, Lipska BK, Ye T, Hyde TM, Tao R, Leek JT, Colantuoni EA, Elkahloun AG, Herman MM, Weinberger DR, Kleinman JE. Temporal Dynamics and Genetic Control of Transcription in the human prefrontal cortex . Nature 2011. Oct 27
Kang HJ, Kawasawa1YI, Cheng F, Zhu Y, Xu X, Li M, Sousa1 AMM, Pletikos M, Meyer KA, Sedmak G, Guennel G, Shin Y, Johnson MB, Krsnik Z, Fertuzinhos MS, Umlauf S, Lisgo SN, Vortmeyer A, Weinberger DR, Mane S, Hyde TM, Huttner A, Reimers M, Kleinman JE, Šestan N. Spatiotemporal transcriptome of the human brain . Nature 2011. Oct 27.
###
The mission of the NIMH is to transform the understanding and treatment of mental illnesses through basic and clinical research, paving the way for prevention, recovery and cure. For more information, visit the NIMH website.
The National Institute on Drug Abuse is a component of the National Institutes of Health, U.S. Department of Health and Human Services. NIDA supports most of the world's research on the health aspects of drug abuse and addiction. The Institute carries out a large variety of programs to inform policy and improve practice. Fact sheets on the health effects of drugs of abuse and information on NIDA research and other activities can be found on the NIDA home page at www.drugabuse.gov. To order publications in English or Spanish, call NIDA's new DrugPubs research dissemination center at 1-877-NIDA-NIH or 240-645-0228 (TDD) or fax or email requests to 240-645-0227 or drugpubs@nida.nih.gov. Online ordering is available at http://drugpubs.drugabuse.gov. NIDA's new media guide can be found at http://drugabuse.gov/mediaguide/.
NINDS is the nation's leading funder of research on the brain and nervous system. The NINDS mission is to reduce the burden of neurological disease — a burden borne by every age group, by every segment of society, by people all over the world.
About the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD): The NICHD sponsors research on development, before and after birth; maternal, child, and family health; reproductive biology and population issues; and medical rehabilitation. For more information, visit the Institute’s Web site at http://www.nichd.nih.gov/ .
About the National Institutes of Health (NIH): NIH, the nation's medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit the NIH website.
The activities described in this release are funded in part through the American Recovery and Reinvestment Act. More information about NIH's Recovery Act grant funding opportunities can be found at http://grants.nih.gov/recovery/ . To track the progress of HHS activities funded through the Recovery Act, visit www.hhs.gov/recovery . To track all federal funds provided through the Recovery Act, visit www.recovery.gov .
