2016 Autumn Inside NIMH

Welcome
Welcome to the latest edition of Inside NIMH! We publish Inside NIMH in conjunction with each meeting of the National Advisory Mental Health Council (NAMHC), which advises the Secretary of Health and Human Services (HHS), the Director of the National Institutes of Health (NIH), and the Director of NIMH on all policies and activities relating to the conduct and support of mental health research, research training, and other programs of the Institute. In addition, check out our website for regular updates on timely topics at NIMH. I hope you find this information interesting and helpful. Please let us know if you have questions or comments on this Autumn 2016 edition.
Sincerely,
Bruce Cuthbert, Ph.D.
Director, RDoC Unit
National Institute of Mental Health
If you wish to unsubscribe, subscribe, or change your email address, please contact the NIMH Webmaster or visit the Inside NIMH subscription page.
Message from the NIMH Director
Just as the leaves on the trees are changing color, NIMH is in transition this autumn. For example, we are pleased to welcome our new Director, Dr. Joshua Gordon! We are also celebrating the completion of the first phase of the Human Connectome Project, the progress of the BRAIN Initiative, and the launch of a new landmark study on adolescent brain development.
As I pass the baton to Dr. Gordon, I am confident that he will lead NIMH in fulfilling its mission to transform the understanding and treatment of mental illnesses. I am gratified to have had the opportunity to lead the Institute over the past several months, and I would like to thank everyone at the Institute for working together to keep NIMH functioning as smoothly as ever during this period. With Dr. Gordon’s arrival, I am resuming the role of Director of the NIMH Research Domain Criteria (RDoC) Unit, and Dr. Shelli Avenevoli is continuing her splendid work as Acting Deputy Director.
Autumn Updates
- NIMH Welcomes New Director, Dr. Joshua Gordon: On July 28, 2016, NIH announced the selection of Joshua A. Gordon, M.D., Ph.D., as the new Director of the NIMH. NIH Director Francis S. Collins, M.D., Ph.D., noted “Josh is a visionary psychiatrist and neuroscientist with deep experience in mental health research and practice. He is exceptionally well qualified to lead the NIMH research agenda to improve mental health and treatments for mental illnesses. We’re thrilled to have him join the NIH leadership team.” Dr. Gordon officially joined NIMH on September 12, 2016. To provide a little background: Dr. Gordon received his degrees from the University of California, San Francisco, where he pioneered the methods necessary to study brain plasticity in the mouse visual system. Before coming to NIMH, Dr. Gordon was an Associate Professor of Psychiatry at Columbia University Medical Center and a Research Psychiatrist at the New York State Psychiatric Institute. His research focuses on the analysis of neural activity in mice carrying mutations of relevance to schizophrenia, anxiety disorders, and depression.
- NIMH Council Workgroup on mHealth Technologies: A National Advisory Mental Health Council (NAMHC) workgroup focusing on Opportunities and Challenges of Developing Information Technologies on Behavioral and Social Science Clinical Research met again on June 13, 2016 to discuss issues related to new mHealth technologies. The group is charged with addressing how these technologies could be used to achieve more objective and precise diagnosis and treatment of mental illnesses, as well as contribute to improved prediction, prevention, and quality in the field of mental health care. The June meeting focused on two key questions: 1) How can these new technologies be used to improve quality in mental health practice? and, 2) How can these new technologies enable new questions to be asked, and enable research to move more rapidly and to become more nimble? The group is now working on a report that will reflect the discussions of this and prior meetings.
- NIMH Answers Questions about Psychosocial Research: NIMH actively supports psychosocial research through a variety of mechanisms, and recently posted a helpful webpage of related questions and answers. Topics include: 1) How NIMH maintains a reasonably balanced portfolio of psychosocial and biological research; 2) How future funding for intervention research could be impacted by NIMH’s experimental therapeutics model for clinical trials; 3) Funding mechanisms for clinical trials; 4) NIMH’s support for treatment process research; 5) Social and community influences on mental health, as well as organizational and structural influences on access to mental health services; 6) Prevention of mental illnesses at all ages; and, 7) NIMH’s role in scientific clinical training and promoting innovative clinical researchers.
- NIH Celebrates Completion of the First Phase of the Human Connectome Project: The NIH Human Connectome Project is an ambitious effort to map the neural pathways that underlie human brain function. On June 20, 2016, NIH celebrated the completion of the Human Connectome Project’s first phase with a public symposium, spotlighting the many successes of the Connectome Project. One such success comes from NIMH grantees Matthew Glasser, Ph.D., and David Van Essen, Ph.D., of Washington University in St. Louis, and colleagues at six other researcher centers, who reported on their discoveries in the journal Nature on July 20, 2016. These researchers used information derived from structural and functional MRI data collected on 210 subjects to create a new 180-region-per-hemisphere map of the cerebral cortex of the human brain. Not only did the researchers map more than double the human cortex’s known regions, but also at the highest resolution to date.
- Future Directions of the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative : The BRAIN Multi-Council Working Group (MCWG) , which works to ensure a coordinated and focused effort on BRAIN across NIH, held their fifth meeting on August 2, 2016 in downtown Washington, DC. The workgroup consists of representatives from the ten NIH Institutes and Centers participating in BRAIN, five at-large members, and ex officio representatives from additional government agencies. Topics discussed included training, technology sharing, tools for cells and circuits, and noninvasive neuromodulation and human brain imaging and recording. The following day, the BRAIN MCWG Neuroethics Workgroup held a meeting to discuss and recommend overall approaches for how research programs like the NIH BRAIN Initiative might handle issues and problems involving ethical issues that can arise from innovative methods of studying the brain.
- NIH Launches Landmark Study on Adolescent Brain Development: NIH recently announced the kick off of the Adolescent Brain Cognitive Development (ABCD) study , which will follow approximately 10,000 children ages 9 to 10 into early adulthood. Scientists will track exposure to substances, academic achievement, cognitive skills, mental health, and brain structure and function using advanced research methods. As the only study of its kind, the ABCD study will yield critical insights into the foundational aspects of adolescence that shape a person’s future. The ABCD study is led by the Collaborative Research on Addiction at NIH (CRAN; National Institute on Drug Abuse, National Institute on Alcohol Abuse and Alcoholism, and National Cancer Institute), and is being conducted in partnership with several institutes, including NIMH.
- Streamlining Multi-site Review of Human Subjects Research: On June 21, 2016, NIH issued the Final NIH Policy on the Use of a Single Institutional Review Board (IRB) for Multi-Site Research (NOT-OD-16-094 ). The new policy enhances and streamlines the IRB review process, setting the expectation that a single IRB of record will be used in the ethical review of non-exempt, NIH-funded, human subjects research protocols that are carried out at more than one site in the United States. This move to a single IRB model also presents a unique opportunity to harmonize the standards and agreements used in clinical research. For more information see the related blog post from the NIH Office of Extramural Research.
- New Research Hospital Board at the NIH Clinical Center: A working group of the Advisory Committee to the NIH Director, called the “Red Team ,” was asked to review multiple aspects of the Clinical Center following the FDA’s finding of significant deficiencies in the Clinical Center pharmacy in May, 2015. One of the Red Team’s recommendations was to establish a new Clinical Center Research Hospital Board . The board met for the first time on July 15, 2016, in an open meeting . Board members noted the importance of engaging the entire community in efforts to improve safety for both patients and employees. Some of the changes already underway include creating the Office of Research Support and Compliance, establishing a search committee for a new chief executive officer to manage the operations of the Clinical Center, and discussing a new governance structure for the Clinical Center. Future meetings and focus groups are being planned to discuss additional changes.
Budget Overview
- Fiscal Year (FY) 2016 Budget: We are currently on track to award approximately 580 new and competing research project grants (RPGs) in FY 2016, an increase from the 531 we estimated for this year. The increase is primarily due to receiving fewer than expected meritorious research centers and research career applications. The projected success rate for competing RPGs in FY 2016 is 22%, an increase from the FY 2015 success rate of 20%. The projected success rate for Early Stage Investigators is 23%, which is consistent with FY 2015. We anticipate funding a total of 83 new Principal Investigators (PIs) in FY 2016, which is consistent with the number of new PIs we funded in FY 2015.
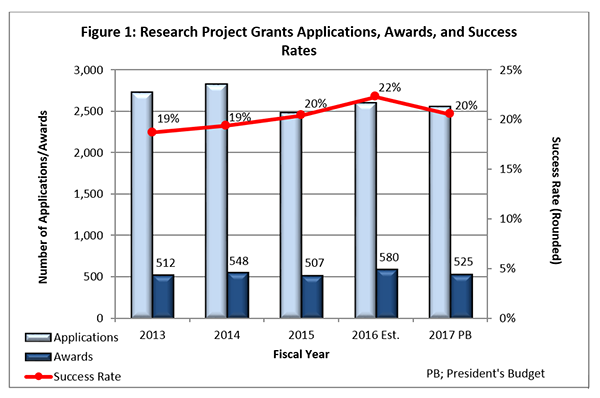
- Outlook for FY 2017: FY 2017 will almost certainly begin under a continuing resolution (CR). As in the past, while operating under a CR, non-competing grants will be awarded at levels below committed amounts, likely at 90%. As in previous years when operating under a CR, the commitment level for NIMH grants will be determined after we receive a full-year appropriation for FY 2017.
NIMH Staff News
- There have been several recent changes to NIMH Leadership.
- Gemma Weiblinger retired from her position as Director of the Office of Constituent Relations and Public Liaison (OCRPL). Ms. Weiblinger first came to NIH from Capitol Hill, working at the National Cancer Institute, and then at the National Institute for Neurological Disease and Stroke, before joining us at NIMH. Over the years she has been key to NIMH’s successful outreach to many Congressional offices and our strong positive relationships with advocacy groups. Ms. Weiblinger was instrumental in efforts to promote understanding about mental illnesses, and the stigma and discrimination affecting those diagnosed with them.
- Phyllis Quartey-Ampofo, M.P.H., is currently serving as Acting Director of the OCRPL. Ms. Ampofo has been with NIMH since 2006, serving as the Institute’s public liaison officer. She has had a lead role executing meetings and interactions with the mental health advocacy and professional organizations, coordinating congressional activities, and facilitating NIMH participation in health education events.
- Tracy Waldeck, Ph.D., Deputy Director of the Division of Extramural Activities, recently moved on to serve as Deputy Director at the Association for Psychological Science. Dr. Waldeck started at NIMH in 2002 as a Staff Psychologist in the Intramural Program under Drs. Manji, Vythilingam, and Drevets, then came to the Extramural Program in 2005 as a Scientific Review Officer, and eventually became Branch Chief of the Extramural Policy Branch. Her breadth of knowledge on extramural grants and grants policies, as well as her expertise in IT systems to support their management, her scientific knowledge, and her collegial work ethic, will be missed.
- Meredith Fox, Ph.D., was officially named Director of the Office of Science Policy, Planning, and Communications (OSPPC). Dr. Fox joined the NIMH Division of Intramural Research Programs (IRP) in 2005, where she studied serotonin and its transporter in the Laboratory of Clinical Science. In 2012, Dr. Fox joined OSPPC, first via a detail and then as a Health Policy Analyst in the Science Policy and Evaluation Branch, rising to Branch Chief in 2014.
- The Division of Intramural Research Programs (IRP) welcomed two new tenure-track investigators in August 2016.
- Soohyun Lee, Ph.D., is Chief of the new Functional Neural Circuits Unit. Her research focuses on how endogenous brain states interact with sensory representation, using the rodent whisker system as a model for studying top-down modulation of sensory processes.
- Argyris Stringaris, M.D., Ph.D., is Chief of the new Mood, Brain, and Development Unit. His research focuses on mood disorders in young people, using a variety of methodological approaches, including psychological and pharmacological interventions.
- Other Staff News:
- Bruno Averbeck, Ph.D., Chief of the Section on Learning and Decision Making in the NIMH IRP, was awarded tenure by the NIH Central Tenure Committee. Dr. Averbeck’s research focuses on understanding the role of frontal-striatal circuits, and particularly the role of dopamine in these circuits, on learning and the representation of beliefs.
Office Spotlight: NIMH Office of Clinical Research
The NIMH Office of Clinical Research (OCR) is involved in many aspects of NIMH-funded clinical research studies, including human subjects protection, data and safety monitoring, recruitment monitoring, regulatory compliance, and certificates of confidentiality. OCR also serves as a liaison between NIMH and other NIH Institutes/Centers, as well as other Federal agencies, for these issues. OCR is comprised of the Human Research Protection Branch and the Clinical Trials Operations and Biostatistics Branch, which has recently implemented the Proactive Site Monitoring Program to assure the highest quality research. OCR has also spearheaded an effort to increase compliance with the registration of clinical trials within ClinicalTrials.gov . This effort has resulted a rate of 97% of all applicable NIMH studies being registered with ClinicalTrials.gov .
OCR Explains: What is a Data and Safety Monitoring Plan and why do I need one? For each NIH-supported clinical trial , NIH requires a data and safety monitoring plan (DSMP) that will provide oversight and monitoring to ensure the safety of participants and the validity and integrity of the data. The level of monitoring should be commensurate with the risks, size, and complexity of the clinical trial. NIMH has developed a helpful resource for investigators creating a DSMP. This and many other resources can be found on the NIMH Clinical Research webpage.
Director’s Highlights: NIMH Scientists and Science
Grantee Awards
NIMH is proud to recognize significant achievements and awards received by our current grantees:
- 2016 Kavli Prize in Neuroscience:
Three NIMH Grantees were named recipients of the prestigious Kavli Prize “for the discovery of mechanisms that allow experience and neural activity to remodel brain function."- Eve Marder, Ph.D. (Brandeis University);
- Michael Merzenich, Ph.D. (University of California, San Francisco); and,
- Carla Shatz, Ph.D. (Stanford University).
Notable NIMH Grants
The following is a selection of the Institute’s most recently funded projects that exemplify our efforts to accelerate research on mental illnesses and to advance the NIMH Strategic Plan for Research:
- Developmental brain disorders (DBDs) comprise a range of conditions, such as autism spectrum disorder, intellectual disability, and schizophrenia, which may share genomic underpinnings. David H. Ledbetter, Ph.D. , and Christa Lese Martin, Ph.D. , (Geisinger Health System) have proposed a genome-first approach to the study of DBDs. These researchers plan to screen 100,000 people with existing genetic data and electronic health records to identify genetic differences that may result in functional differences in traits like cognitive performance, adaptive behavior, and social function. This project may identify new DBD genes and help researchers better understand the phenotypic spectrum associated with DBD gene expression, as well as penetrance (the likelihood that expressing a gene will result in an associated trait). These findings may provide insight into the range of effects of deleterious DBD genes in the general population, ultimately influencing diagnostic as well as treatment practices.
- Lack of motivation is a symptom that impairs older adolescents and adults with attention deficit hyperactivity disorder (ADHD), leading to high disease burden (e.g., school drop-out and poor job performance). Rachel Adcock, M.D. , and Scott Kollins, Ph.D. (Duke University) propose using a novel intervention targeting motivation to treat patients with ADHD. They have already shown that healthy volunteers are able to self-stimulate the ventral tegmental area (VTA), part of the brain circuitry underlying reward that is dysregulated in ADHD, by imagining personalized scenarios to increase motivation; now, they plan to test this ability in adults with ADHD. These researchers aim to evaluate whether activity can be enhanced by providing patient feedback using real-time fMRI, and whether self-stimulation is associated with improvement in motivation, cognition, and ADHD symptoms. If individuals with ADHD can self-stimulate the VTA, this could be the basis for a non-drug, individually tailored treatment to enhance motivation and clinical functioning in patients with ADHD.
- Although cognitive therapy (CT) is well-established as an effective treatment for major depression, in actual practice, patient response is often uneven. Allison G. Harvey, Ph.D. (University of California at Berkeley) will conduct a study, “Improving Depression Outcome by Enhancing Memory for Cognitive Therapy,” which targets a common variable that has been shown to mediate response to CT – the ability to remember the actual contents of each therapy session. This confirmatory efficacy trial aims to enhance routine CT for depression by employing memory-promoting strategies (“Memory Support Intervention”) as an integral component of the therapy to improve the often-poor memory for the contents of a therapy session – a risk factor for diminished adherence to treatment and clinical outcome. This novel, cost-effective, easily scalable, and generalizable memory enhancement intervention holds the potential for increasing the effectiveness of CT in depression (and perhaps other disorders).
For more information on these and other grants selected for funding, please visit the NIH RePORTER website .
Current Funding Opportunities and Announcements
NIH electronically posts the NIH Guide , a listing of all NIH Funding Opportunity Announcements (FOAs), including requests for applications (RFAs), program announcements (PAs), and important notices for the scientific community. Below is a selection of recently issued FOAs in which NIMH participates. The Funding page on the NIMH website has links to listings of all NIMH FOAs and other resources.
You can subscribe to the NIMH Funding Opportunities ListServ to receive the latest information about RFAs and other research funding opportunities from NIMH, as well as administrative updates and changes to grant policies and procedures. You can also subscribe to a separate listserv to receive weekly e-mails of the NIH Guide .
NIMH-Administered Requests for Applications
- Exploratory Clinical Trials of Novel Interventions for Mental Disorders
- Release date: March 30 and May 22, 2015; Application due dates: October 14, 2016
- R33 announcement (RFA-MH-16-400 )
- R61/R33 announcement (RFA-MH-16-406 )
- Pilot Effectiveness Trials for Treatment, Preventive and Services Interventions
- Release date: March 30, 2015; Application due dates: October 14, 2016
- R34 announcement (RFA-MH-16-410 )
- Clinical Trials to Test the Effectiveness of Treatment, Preventive and Services Interventions
- Release date: March 30, 2015; Application due dates: October 14, 2016
- Collaborative R01 announcement (RFA-MH-16-415 )
- R01 announcement (RFA-MH-16-420 )
- Confirmatory Efficacy Clinical Trials of Non-Pharmacological Interventions for Mental Disorders
- Release date: March 30, 2015; Application due dates: October 14, 2016
- R01 announcement (RFA-MH-16-425 )
- NIMH Biobehavioral Research Awards for Innovative New Scientists (NIMH BRAINS)
- Release date: June 11, 2014; Application due date: October 24, 2016
- R01 announcement (RFA-MH-15-600 )
- Applied Research Toward Zero Suicide Healthcare Systems
- Release date: December 11, 2015; Application due date: November 2, 2016
- R01 announcement (RFA-MH-16-800 )
- BRAIN Initiative: Development and Validation of Novel Tools to Analyze Cell-Specific and Circuit-Specific Processes in the Brain
- Release date: August 10, 2016; Application due date: November 2, 2016 and October 13, 2017
- R01 announcement (RFA-MH-17-220 )
- BRAIN Initiative: Foundations of Non-Invasive Functional Human Brain Imaging and Recording - Bridging Scales and Modalities
- Release date: August 15, 2016; Application due date: November 23, 2016 and October 13, 2017
- R01 announcement (RFA-MH-17-235 )
- BRAIN Initiative: Non-Invasive Neuromodulation - Mechanisms and Dose/Response Relationships for Targeted CNS Effects
- Release date: August 23, 2016; Application due date: November 23, 2016 and October 13, 2017
- R01 announcement (RFA-MH-17-245 )
- BRAIN Initiative: Non-Invasive Neuromodulation - New Tools and Techniques for Spatiotemporal Precision
- Release date: August 23, 2016; Application due date: November 23, 2016 and October 13, 2017
- R01 announcement (RFA-MH-17-240 )
- Understanding and Addressing the Multi-level Influences on Uptake and Adherence to HIV Prevention Strategies Among Adolescent Girls and Young Women in Sub-Saharan Africa
- Release date: August 15, 2016; Application due date: December 20, 2016
- R01 announcement (RFA-MH-17-550 )
- R21 announcement (RFA-MH-17-555 )
- R34 announcement (RFA-MH-17-560 )
- Using the NIMH Research Domain Criteria (RDoC) Approach to Understand Psychosis
- Release date: March 17, 2016; Standard due dates apply: October 5, 2016 – March 5, 2017
- R01 announcement (PAR-16-136 )
- R21 announcement (PAR-16-135 )
- Reducing the Duration of Untreated Psychosis in the United States
- Release date: May 17, 2016; Standard due dates apply: November 18, 2016 – March 19, 2019
- R01 announcement (PAR-16-265 )
- R34 announcement (PAR-16-264 )
NIMH-Collaborative Requests for Applications
- Big Data to Knowledge (BD2K) Community-based Data and Metadata Standards Efforts
- Release date: August 16, 2016; Application due dates: October 19, 2016; October 19, 2017
- R24 announcement (RFA-ES-16-010 )
- Limited Competition for the Continuation of the National Consortium on Alcohol and Neurodevelopment in Adolescence (NCANDA)
- Release date: May 31, 2016; Application due date: November 9, 2016
- Research Project Sites U01 announcement (RFA-AA-17-003 )
- Administrative Resource U24 announcement (RFA-AA-17-004 )
- Data Analysis Resource U24 announcement (RFA-AA-17-005 )
- NIH Big Data to Knowledge (BD2K) Enhancing Diversity in Biomedical Data Science
- Release date: August 8, 2016; Application due date: November 14, 2016
- R25 announcement (RFA-MD-16-002 )
- Autism Centers of Excellence: Centers and Networks
- Release date: July 12, 2016; Application due date: November 17, 2016
- R01 announcement (RFA-HD-17-008 )
- P50 announcement (RFA-HD-17-009 )
- Limited Interaction Targeted Epidemiology (LITE) to Advance HIV Prevention
- Release date: May 26, 2016; Application due date: December 5, 2016
- UG3/UH3 announcement (RFA-AI-16-031 )
- Silencing of HIV-1 Proviruses
- Release date: May 26, 2016; Application due date: December 7, 2016
- R61/R33 announcement (RFA-AI-16-038 )
- BD2K Research Education Curriculum Development: Data Science Overview for Biomedical Scientists
- Release date: August 10, 2016; Application due dates: December 7, 2016; December 1, 2017
- R25 announcement (RFA-ES-16-011 )
- Big Data to Knowledge (BD2K) Enhancing the Efficiency and Effectiveness of Digital Curation for Biomedical Big Data
- Release date: August 24, 2016; Application due date: December 15, 2016
- U01 announcement (RFA-LM-17-001 )
- BD2K Open Educational Resources for Skills Development in Biomedical Big Data Science
- Release date: May 18, 2016; Application due date: August 2, 2017
- R25 announcement (RFA-HG-16-016 )
- Understanding HIV Persistence in Infants
- Release date: August 5, 2016; Application due date: December 7, 2017
- R01 announcement (RFA-AI-16-064 )
- Rapid Assessment of Zika Virus (ZIKV) Complications
- Release date: February 19, 2016; Application due date: Rolling
- R21 announcement (PAR-16-106 )
Future Research Directions
Concept Clearances for Potential New Research Initiatives
This listing of potential future initiatives is meant to provide the earliest possible alert to the field of our research interests and of potential upcoming announcements to solicit that research. While NIMH plans to proceed with these initiatives, their publication and timing are not certain and depend on sufficient funding. The titles and brief descriptions are consistent with the information available at the time of concept clearance. The resultant FOAs may differ from the concepts in the final wording of their titles or other aspects. To send questions about a specific concept, follow the “Submit Comments” link at the bottom of the description.
- Addressing Suicide Research Gaps: Understanding Mortality Outcomes
- Addressing Suicide Research Gaps: Aggregating Existing Data Sets for Secondary Analyses
- Transdisciplinary Research Centers to Advance Clinical Practice in Mental Health
- Limited Competition for Renewal of the National NeuroAIDS Tissue Consortium
- Personalized Mental Health Navigator to Promote Early Engagement, Linkage and Coordination of Needed Mental Health Services for Children and Adolescents
- NIMH Center for Genomic Research Resources for Advancing Psychiatric Genetics
- Research to Support the Reduction and Elimination of Mental Health Disparities
- NIMH Biobehavioral Research Awards for Innovative New Scientists (BRAINS)
For more information, please see recent NAMHC-approved concepts, recent public venue-approved concepts, and past NAMHC meetings, which also contains links to meeting agendas, minutes, and Inside NIMH (Director’s Reports).
NIMH-Sponsored Meetings
- Mechanisms Underlying Suicide Risk: Integrating RDoC to Inform Novel and Personalized Intervention Research: On June 2-3, 2016, the Division of Translational Research (DTR) and the Division of Services and Intervention Research (DSIR) sponsored a workshop to inform personalized treatment development by identifying potential intervention constructs for those at risk for suicidality. With input from the National Institute on Drug Abuse , the Eunice Kennedy Shriver National Institute of Child Health and Human Development , and the National Institute on Alcohol Abuse and Alcoholism , a multidisciplinary group of researchers, representing expertise in development, neuroimaging, neural circuitry, pathophysiology, behavior, risk, and intervention, convened to review current themes in suicide research, and to consider how research approaches consistent with the RDoC framework could provide new insight on mechanisms underlying suicide risk.
- Global HIV-1 CSF Escape Consortium: On June 9, 2016, the Division of AIDS Research, with support from the NIH Office of AIDS Research , convened a meeting entitled “Global HIV-1 CSF Escape Consortium.” Effective antiretroviral therapy has changed the nature of living with HIV, such that sustained treatment can result in complete viral suppression. However, several clinical studies have demonstrated that despite stable and successful control of HIV in the periphery, some patients still have detectable virus in their cerebrospinal fluid (CSF), termed “CSF escape.” Meeting participants identified key scientific research priorities and outlined steps to establish a common database to catalogue cases and facilitate collaborations.
- 2016 Autism Centers of Excellence (ACE) Investigator Meeting: On June 13-14, 2016, extramural investigators from the three centers and nine networks that comprise the NIH Autism Centers of Excellence Program convened for their annual meeting at the NIH Neuroscience Center. The meeting provided ACE investigators an opportunity to share the progress of their respective centers and networks, as well as to discuss current challenges and promising innovations that may advance autism research. Meeting topics included the impact of epilepsy on developmental testing among infants affected by autism, the advantages of utilizing adaptive interventions in studies of autism, the strength of new targets and strategies for interventions, and new approaches to measuring and quantifying the development of infants affected by ASD, at home and in research settings.
- Research on Early Identification and Linkage to Services for ASD: On June 15, 2016, DSIR’s Services Research and Clinical Epidemiology Branch convened a meeting with researchers supported by the NIMH funding opportunity “Services Research for Autism Spectrum Disorder across the Lifespan: Research on Early Identification and Linkage to Services for ASD .” This meeting focused on the identification of potential strategies to leverage their collective data to address additional scientific areas of inquiry. The five studies funded from this opportunity have formed the NIMH ASD Prevention, Early Detection, Engagement and Services (ASD PEDS) Research Network, and are testing strategies for universal ASD screening and early referral to services for children ages 0 to 3. The Network is also collecting data using common measures and plans to pool data to conduct additional analyses.
- Outreach Partnership Program (OPP): On July 19-21, 2016, the Office of Constituency Relations and Public Liaison convened the NIMH OPP Annual Meeting in Bethesda, MD. Acting NIMH Director Dr. Bruce Cuthbert provided an update on the state of mental health research to the 55 NIMH Outreach Partners and some of the more than 80 National Partner organizations. Other presentations highlighted NIMH psychosis efforts, suicide prevention strategies, the science of mindfulness, advancements in the treatment of refractory depression, improving access to care to reduce mental health disparities, and the use of trauma-focused cognitive behavioral therapy for children and adolescents. Participants also learned about activities carried out by select Outreach Partners to train the criminal justice and law enforcement community to understand and address the mental health impacts of trauma from domestic violence.
- NIMH Conference on Mental Health Services Research (MHSR): On August 1-2, 2016, NIMH hosted the 23rd MHSR meeting. This year’s theme was “Harnessing Science to Strengthen the Public Health Impact.” The conference brought together hundreds of leading mental health services researchers, clinicians, advocates, and federal and nonfederal partners. Services research aims to improve access to mental health care, as well as improve continuity, quality, equity, and value of care, a mission crucial to closing the science to service delivery gap. The conference highlighted the essential role of mental health services research in improving the efficiency and effectiveness of mental health services, establishing research partnerships, developing innovative service delivery models, and evaluating the public health impact of mental health research.
- 2016 Webinar Series on Mental Health Disparities and Global Mental Health Research: The Office for Research on Disparities and Global Mental Health organized two webinar series that ran from May through August, 2016.
- The series on mental health disparities included three webinars. The first webinar, “Building Resilience to Reduce Suicide in Arctic Communities,” highlighted innovative community-based research on interventions designed to reduce youth suicide in Arctic communities. The second webinar, “Translational Research in Women’s Mental Health,” featured research on reproductive hormone-related mood disorders, and the phenotypic and genetic signature of perinatal mood disorders. The final webinar focused on “Mental Health Disparities Research and the NIMH Strategic Plan.”
- The series on global mental health research included four webinars. The first webinar, “How to Manage Your NIMH Grant: Guidance for Low- or Middle-Income Countries,” featured grants management specialists sharing tips for grantees. "Getting the Most from Your Data: Sharing it through the NIMH Data Archive,” described depositing and accessing data in the NIMH Data Archive. The third webinar, “Good Clinical Practice for Conducting Clinical Trials Research in Low- or Middle-Income Countries,” introduced standards for conducting clinical trials research worldwide. The final webinar was titled “Implementation Science: An NIH Perspective on Implementation Research in Low- and Middle-Income Countries.”
Update on Electronic Research Administration (eRA) Activities
Electronic Grant Application Submission News
- Use Application FORMS-D Posted with FOAs despite Expiration Dates on R&R Forms: NIH grant application form packages include both agency-specific forms (typically labeled PHS) and federal-wide forms (typically labeled Research & Related, or R&R). Although agency-specific forms were recently approved for use through October 31, 2018, the forms used federal-wide are on different clearance cycles and have recently expired or will expire soon. Applicants and grantees should continue to use the application form packages (FORMS-D) posted with funding opportunity announcements despite the expiration dates noted on individual forms. Proposed updates to the federal-wide forms should have minimal user impact (NOT-OD-16-120 ).
NIH-Wide Grant News
- The NIH Regional Seminar on Program Funding and Grants Administration will be held in Chicago, IL, on October 26-28, 2016. If you or your staff are new to working with NIH grants, then this is an opportunity to meet with and get questions answered by NIH staff. Here are just a few highlights:
- Pre-seminar workshops on research with human participants, intellectual property, electronic Research Administration (eRA) systems, and the NIH grants process;
- Presentations will cover over 40 different funding and grants administration topics; and,
- 1:1 sessions with NIH and HHS presenters.
- NIH Extramural Loan Repayment Programs (LRP) Announced: There are some changes to the LRP application process, including that applicants are now required to be registered in eRA Commons. The deadline for LRP submissions is November 12, 2016 (NOT-OD-16-119 , NOT-OD-16-118 , NOT-OD-16-117 , NOT-OD-16-116 , and NOT-OD-16-115 ).
For more information on all of these updates, please see the NIH eRA News and Events page .
Questions? Contact the eRA help desk . Note that contacting this help desk is the only way to document problems with an electronic grant application submission. Evidence of this contact is the only way to be eligible for any special consideration by the Center for Scientific Review (CSR) Division of Receipt and Referral, should you run into a system problem with Grants.gov or with eRA that is beyond your control.
Research Training and Career Development
Here is the latest news about Research Training and Career Development at the NIMH and the NIH:
- We encourage applications for our three administrative supplement programs that provide funds to NIMH research project grants to support research experiences for individuals early in their careers:
- Research Supplements to Promote Diversity in Health-Related Research ;
- NIMH Administrative Supplement Program to Enable Continuity of Research Experiences of MD/PhDs during Clinical Training ; and,
- NIMH Administrative Supplement Providing Research Experiences for Physicians and Medical Students from Diverse Backgrounds .
- New Investigators Workshop: On August 3, 2016, in conjunction with the MHSR Conference, the Services Research and Clinical Epidemiology Branch of the NIMH Division of Services and Intervention Research held a New Investigators Workshop. During the workshop, 24 competitively-selected attendees heard presentations relevant to research training and career development, how to navigate the NIMH grant application process, and current Institute priorities in mental health services research. Attendees also presented their completed research projects at the MHSR poster session and discussed their research ideas with workshop faculty and peers during small group break-out sessions.
Please refer to the NIMH webpage for research training and career development between issues of Inside NIMH to locate the latest news and resources for potential applicants and current awardees.
We are interested in feedback from the community; comments or suggestions related to NIMH’s support for research training and career development may be directed to NIMH_Training@mail.nih.gov.
NIMH Science Updates
The latest news and updates from NIMH-supported research:
- NIMH Funds 3 ‘Zero Suicide’ Grants (September 16, 2016)
- Circuitry for Fearful Feelings, Behavior Untangled in Anxiety Disorders (September 9, 2016)
- Designer Agent Blocks Pain in Mice Without Morphine’s Side Effects (August 17, 2016)
- How “Quickly Forgotten” Early Life Experiences Mature the Brain (August 15, 2016)
- Schizophrenia, Autism Risk Gene Trajectories Point to Shared Causes (August 12, 2016)
- NIMH Grantees Named Recipients of Prestigious Kavli Prize (August 3, 2016)
- Coping With Familial Mental Illness in Stressful Times (July 28, 2016)
- Electroconvulsive Therapy Lifts Depression, Sustains Remission in Older Adults (July 18, 2016)
- Game Corrects Children’s Misreading of Emotional Faces to Tame Irritability (June 22, 2016)
Publicizing NIMH research is a communal responsibility. Please help us spread the word about the results of NIMH funding by acknowledging our support of your research, for example, in journal articles (citing your NIMH award by number when possible) and other communications. NIMH has two primary methods of getting the word out: press releases and science updates. All releases and updates are posted to the Science News section of the NIMH Web site. These are also distributed to the public through a mailing list .
Connect with NIMH
Our newest effort to reach our stakeholders is a service that allows you to subscribe for updates sent directly to your email inbox on the NIMH topics of your choice. In addition to our email newsletters and RSS updates, please also visit NIMH on Twitter , Facebook , and YouTube , where we highlight Science Updates, Press Releases, and other timely matters.
Inside NIMH is produced by the National Institute of Mental Health. For more information about the Institute, visit our website at https://www.nimh.nih.gov. For comments and suggestions about Inside NIMH, please contact the NIMH Webmaster. The material in this newsletter is not copyrighted, and we encourage its use or reprinting.
