Archived Content
The National Institute of Mental Health archives materials that are over 4 years old and no longer being updated. The content on this page is provided for historical reference purposes only and may not reflect current knowledge or information.
Estrogen Alters Memory Circuit Function in Women with Gene Variant
Hormone-gene interaction may underlie sex/individual differences in mental disorders
• Science Update
Fluctuations in estrogen can trigger atypical functioning in a key brain memory circuit in women with a common version of a gene, NIMH scientists have discovered. Brain scans revealed altered circuit activity linked to changes in the sex hormone in women with the gene variant while they performed a working memory task.
The findings may help to explain individual differences in menstrual cycle and reproductive-related mental disorders linked to fluctuations in the hormone. They may also shed light on mechanisms underlying sex-related differences in onset, severity, and course of mood and anxiety disorders and schizophrenia. The gene-by-hormone interaction’s effect on circuit function was found only with one of two versions of the gene that occurs in about a fourth of white women.
Drs. Karen Berman, Peter Schmidt, Shau-Ming Wei, and colleagues, of the NIMH Intramural Research Program, report on this first such demonstration in women April 18, 2017 in the journal Molecular Psychiatry.
Prior to the study, there was little evidence from research on the human brain that might account for individual differences in cognitive and behavioral effects of sex hormones. For example, why do some women develop postpartum depression and others do not – in response to the same hormone changes? Why do some women report that estrogen replacement improved their memory, whereas large studies of postmenopausal estrogen therapy show no overall improvement in memory performance?
Evidence from humans has also been lacking for the neural basis of stark sex differences in prevalence and course of mental disorders that are likely related to sex hormones. For example, why are there higher rates of mood disorders in females and higher rates of ADHD in males – or later onset of schizophrenia in females?
In seeking answers to these questions, the researchers focused on working memory, a well-researched brain function often disturbed in many of these disorders. It was known that working memory is mediated by a circuit from the brain’s executive hub, the prefrontal cortex, to its memory hub, the hippocampus. Notably, hippocampus activity is typically suppressed during working memory processing.
Following-up on a clue from experiments in mice, the NIMH team hypothesized that estrogen tweaks circuit function by interacting with a uniquely human version of the gene that codes for brain derived neurotrophic factor (BDNF), a pivotal chemical messenger operating in this circuit. To find out, the researchers experimentally manipulated estrogen levels in healthy women with one or the other version of the BDNF gene over a period of months. Researchers periodically scanned the women’s brain activity while they performed a working memory task to see any effects of the gene-hormone interaction on circuit function.
The researchers first scanned 39 women using PET (positron emission tomography) and later confirmed the results in 27 women using fMRI (functional magnetic resonance imaging). Both pegged atypical activity in the hippocampus to the interaction. Turning up the same findings using two types of neuroimaging strengthens the case for the accuracy of their observations, say the researchers. Such gene-hormone interactions affecting thinking and behavior are consistent with findings from animal studies and are suspect mechanisms conferring risk for mental illness, they add.
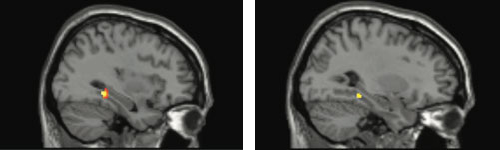
activation (yellow) in the brain’s memory hub, or hippocampus, in
response to estrogen in women performing a working memory task
– if they carried a uniquely human version of the BDNF gene. Activity
in this area is typically suppressed during working memory. Picture
shows PET and fMRI data superimposed over anatomical MRI image.

NIMH Section on Integrative
Neuroimaging, was first
author of the study.
Reference
Brain-derived neurotrophic factor Val66Met genotype and ovarian steroids interactively modulate working memory-related hippocampal function in women: a multimodal neuroimaging study. Wei SM, Baller EB, Kohn PD, Kippenhan JS, Kolachana B, Soldin SJ, Rubinow DR, Schmidt PJ, Berman KF.Mol Psychiatry. 2017 Apr 18. doi: 10.1038/mp.2017.72. [Epub ahead of print] PMID:28416813
