2017 Winter Inside NIMH

Welcome
Welcome to the latest edition of Inside NIMH! We publish Inside NIMH in conjunction with each meeting of the National Advisory Mental Health Council, which advises the Secretary of Health and Human Services (HHS), the Director of the National Institutes of Health (NIH), and the Director of NIMH on all policies and activities relating to the conduct and support of mental health research, research training, and other programs of the Institute. In addition, check out our website for regular updates on timely topics at NIMH. I hope you find this information interesting and helpful. Please let us know if you have questions or comments on this edition.
Sincerely,
Joshua A. Gordon, M.D., Ph.D.
Director, National Institute of Mental Health
If you wish to unsubscribe, subscribe, or change your email address, please contact the NIMH Webmaster or visit the Inside NIMH subscription page .
NIMH Director’s Updates
As the new President settles into the Oval Office, I too am settling in as the Director at NIMH. While I continue to listen and learn, I have also identified some priorities for the Institute.
Presidents and Priorities
- Changes in Leadership: With the New Year comes new opportunity, a new political landscape, and continued commitment to transform mental health care. I look forward to working with the incoming Presidential Administration to ensure that the outstanding efforts of NIMH staff and funding recipients continue to be recognized and appreciated, and that improving mental health care through research remains a priority. I am sincerely grateful to the leadership, staff, grantees, and larger NIMH community for working with me these past few months. They have welcomed me with open minds, new ideas, and patience. I would like to especially thank Bruce Cuthbert, Ph.D., for his exemplary and dedicated leadership while serving as the NIMH Acting Director, and Shelli Avenevoli, Ph.D., who is continuing on as NIMH Deputy Director.
- Listening and Learning: In order to understand what the major issues are, and where there are gaps in knowledge and gaps in our research efforts, I intend to spend most of my first year at NIMH listening and learning. I will also continue to communicate my thoughts with the broad NIMH constituency through Director’s Messages. My first message, Freshman Year, focused on diversity as the cornerstone of a strong research portfolio – diversity in subject matter, diversity in the research workforce and study participants, and diversity in time frames. Specifically, NIMH should support studies that will yield benefits on short-, medium-, and long-term timescales. I also discussed three of my research priorities: suicide prevention, neural circuits, and computational and theoretical approaches to psychiatry. In my second Director’s Message, The Push for Suicide Prevention, I outlined NIMH’s current efforts, gaps in the research, and possible approaches to suicide prevention. Research in suicide prevention has the potential to yield benefits in the short-term. In Neural Circuits Research: How and Why, my third Director’s message, I described an approach to translating neural circuit technology into novel treatment methods. This approach has the potential to yield benefits in the medium-term. Right now, our understanding of which circuits underlie certain features of mental disorders is rudimentary at best. One goal is to identify behaviors that are disruptive, and then manipulate the underlying circuits to reduce a person's disability without altering his or her abilities. Scientists can then develop treatments that aim at fine-tuning the wiring that underlies the circuitry. Finally, I believe that further development of computational and theoretical approaches to psychiatry has high potential impact. For example, computational modeling that incorporates clinical data to build predictive biomarkers or a framework for diagnosis could truly transform mental health care. An upcoming Director’s Message will focus on these approaches.
- Updates on Ongoing NIMH and NIH Initiatives:
- National Advisory Mental Health Council (NAMHC) Workgroup Updates: Since the last Council meeting, three NAMHC workgroups have been busy. The NAMHC workgroup on the Opportunities and Challenges of Developing Information Technologies on Behavioral and Social Science Clinical Research is finalizing a report that summarizes their discussions on new mHealth technologies. Two NAMHC workgroups recently convened to address different aspects of the Research Domain Criteria (RDoC) initiative. The first workgroup was tasked with identifying optimal behavioral tasks and measures to assess RDoC constructs. A report of their August 2016 discussions is now available, and highlights existing tools and areas for future task development. The second workgroup is tasked with evaluating proposed changes to the RDoC matrix. On November 3-4, 2016, they cohosted a Motor Systems Workshop with the NIMH RDoC unit. The purpose of the workshop was to develop consensus and provide advice to NIMH regarding the addition of a motor domain to the RDoC matrix. Additionally, a new genomics workgroup will advise the NAMHC on future directions in psychiatric genetics and functional genomics, including how best to address the gap in knowledge between gene discovery and mechanistic models of disease that transcend categorical DSM disease classification.
- Research Domain Criteria (RDoC) “Office Hours”: Are you a researcher with questions about the RDoC initiative? The members of the NIMH RDoC Unit are holding monthly virtual “office hours.” Topics that may be addressed during the office hours include description of the basic principles of the initiative, general queries about designing studies and applying for grants based on RDoC principles, and the role of RDoC in relation to the general NIMH Strategic Research Priorities and other initiatives. Office hours will be held via video conference on the second Friday of every month.
- NIMH Released New Clinical Trials Funding Opportunity Announcements (FOAs): On December 13, 2016, NIMH issued eight new clinical trial-specific FOAs to support clinical trial applications. NIMH requires an experimental therapeutics approach to the development and testing of interventions, in which interventions serve not only as potential treatments, but as probes to generate information about the mechanisms underlying a disorder. Changes were made to all clinical trials FOAs in an effort to clarify NIMH’s interest in this experimental therapeutics approach to the development and testing of interventions across all modalities. One major change is that there are now two separate “early stage” FOAs for the development of 1) psychosocial interventions, and 2) drugs or direct brain modulation/stimulation devices. Applicants should carefully read the FOA to which they are responding, and are strongly encouraged to contact the Scientific/Research Contact listed at the end of the relevant FOA to discuss the FOA and NIMH priorities. Also see the Applicant FAQs.
- NIH Nearly Doubled Investment in Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative : In October 2016, NIH announced its third round of grants to support the goals of the BRAIN Initiative. This latest round of grants includes over one hundred new awards, totaling more than $70 million, and will go to over 170 investigators working at 60 institutions. These grants bring NIH’s total fiscal year 2016 investment in BRAIN to just over $150 million and expand NIH’s efforts to develop new tools and technologies to understand neural circuit function and capture a dynamic view of the brain in action. Some NIMH-administered BRAIN projects are aimed at fine-tuning brain stimulation and other promising technologies for the treatment of mental illnesses.
- 21st Century Cures Act Impact on Mental Health: The 21st Century Cures Act was signed into law on December 13, 2016. Overall, the mental health and substance use provisions aim to strengthen leadership and accountability at the Substance Abuse and Mental Health Administration; increase collaboration and consultation to promote the implementation of evidence-based programs; continue support for state programs; and, provide assistance to individuals, families, and providers in accessing care and resources. The law authorizes spending $4.8 billion over ten years for the Cancer Moonshot Initiative , Precision Medicine Initiative , BRAIN Initiative , and Regenerative Medicine Program. Additional provisions relate to primary-behavioral health care integration, parity, HIPAA compliance and communication of health information, training and information dissemination, community crisis response and intervention, and guidance concerning interactions with the judicial system. Finally, the 21st Century Cures Act aims to reduce regulations and administrative burden on researchers, and includes provisions to reinforce the importance of including diverse populations in research.
- Landmark “Common Rule” Finalized: The U.S. Department of Health and Human Services and fifteen other Federal Departments and Agencies issued final revisions to the Federal Policy for the Protection of Human Subjects, also known as the “Common Rule.” This revised policy aims to better protect human subjects involved in research; facilitate valuable research; and, reduce burden, delay, and ambiguity for research investigators. The final Common Rule was published in the Federal Register on January 19, 2017.
Budget Overview
- Fiscal Year (FY) 2016 Budget: NIMH awarded an estimated 587 new and competing research project grants (RPGs) in 2016, and achieved an overall success rate of approximately 23 percent (defined as number of RPG applications funded divided by the number of applications received; see Figures 1 and 2 below). NIMH awarded grants to 84 New Investigators, and achieved a success rate of approximately 24 percent for Early Stage Investigators .
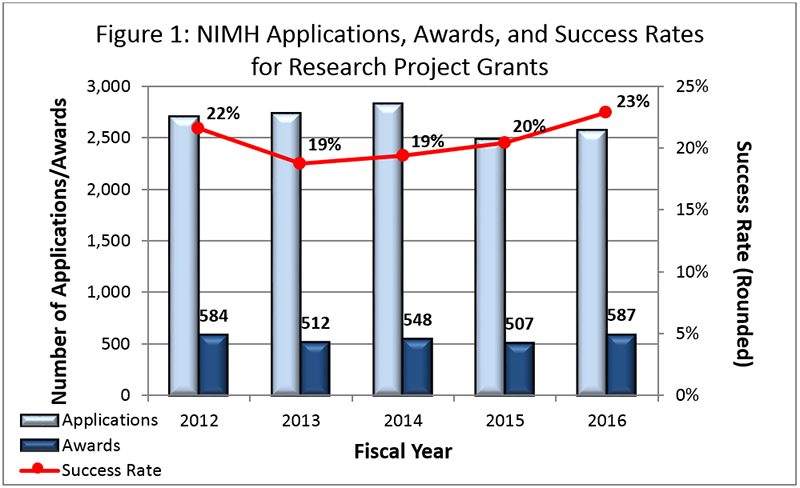
Figure 2 below shows the number of applications received and the number of awards made through the 25th percentile for RPGs. Not included are applications/awards higher than the 25th percentile. Note that in Figure 2, the total number of funded grants do not add up to 587, as not all grants are percentiled.
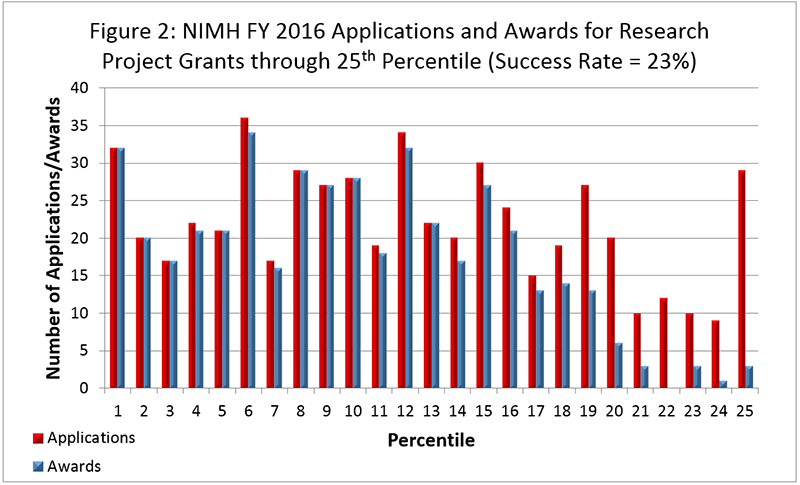
Figure 3 below shows the NIMH budget in appropriated (current) versus constant (FY 1998) dollars. Constant dollars are “inflation adjusted” for variations in the purchasing power of the dollar over time. Dollar amounts are adjusted based on the Biomedical Research and Development Price Index (BRDPI). The annual change in BRDPI indicates how much the NIH budget must change to maintain purchasing power similar to FY 1998.
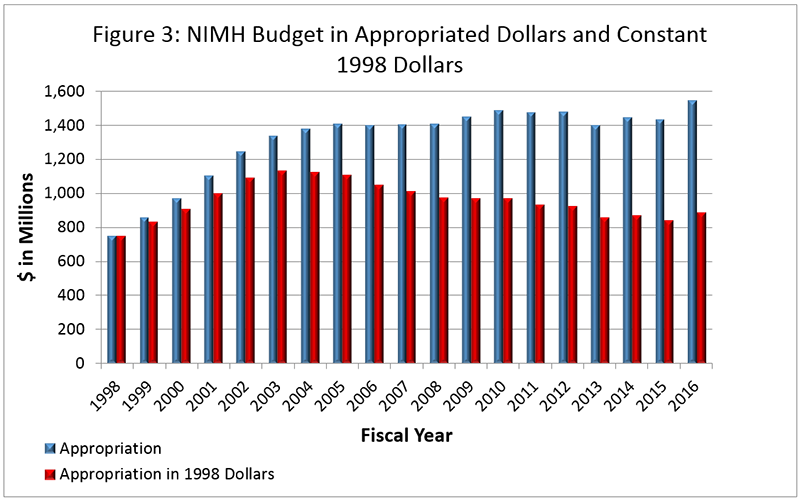
- FY 2017 Budget: Although FY 2017 began on October 1, 2016, there is still uncertainty regarding the NIH and NIMH budgets. In order to continue operations, a continuing resolution (CR) was signed by President Obama on September 29, 2016 to provide funding through December 9, 2016. A second CR was signed on December 10, 2016 to continue funding through April 28, 2017. Funds for the 21st Century Cures Act were provided under this latest CR in the amount of $352 million in a new NIH Innovation Account. Funds have been provided for the following Initiatives for FY 2017: Cancer Moonshot ($300,000,000), Precision Medicine Initiative ($40,000,000), BRAIN Initiative ($10,000,000), and Regenerative Medicine ($2,000,000).
While NIMH continues to operate under a CR, the Institute is issuing non-competing research grant awards at a level lower than indicated on the most recent Notices of Award (generally up to 90 percent of the previously committed level). As in previous CRs, we look forward to upward adjustments after the final appropriation is enacted later in the fiscal year.
NIMH Staff News and Awards
- There have been several recent changes to NIMH Leadership.
- Shelli Avenevoli, Ph.D., was officially named NIMH Deputy Director. Dr. Avenevoli served as the Institute’s Acting Deputy Director since October 2015. In 2001, she joined NIMH as a staff scientist in the intramural research program. In 2005, Dr. Avenevoli moved to the NIMH division of extramural research as Chief of the Emotion, Mood, and Depressive Disorders Program, and became Chief of the Developmental Trajectories of Mental Disorders Branch in 2008. She has been heavily involved in a number of key NIMH efforts, including revising NIMH’s Strategic Plan for Research, re-defining the Institute’s approach to supporting research in neurodevelopment and bipolar disorder, and serving as a liaison to other agencies for special initiatives. Dr. Avenevoli received her Ph.D. in Developmental Psychology from Temple University.
- Bruce Cuthbert, Ph.D., Director of the NIMH Research Domain Criteria (RDoC) Unit, formally retired at the end of November 2016. He will remain involved with NIMH, and particularly the work of the RDoC Unit. Dr. Cuthbert has held a number of leadership positions at NIMH, most recently serving as the NIMH Acting Director from 2015 to 2016 and as the Director of the Division of Adult Translational Research from 2009 to 2014. He also served as Chief of the Emotion Process Program, Acting Chief of the Biobehavioral Regulation Program, and Chief of the Adult Psychopathology and Prevention Research Branch while in the NIMH extramural program from 1998 to 2005. He left NIMH in 2005 to join the University of Minnesota as a professor of Clinical Psychology, returning to NIMH in 2009 to take on the coordination of RDoC. Dr. Cuthbert received his Ph.D. in Clinical Psychology from the University of Wisconsin–Madison.
- Meena Hiremath, Ph.D., P.M.P., is the new Deputy Director for the Division of Extramural Activities (DEA). Dr. Hiremath comes to NIMH from the National Heart, Lung, Blood Institute where she served as Director of the Office of Extramural Policy and Training. In that role, she oversaw the coordination of extramural operations, such as Council and initiative development, and staff training. She began her extramural career as a Scientific Program Analyst at the National Institute of Neurological Disorders and Stroke and worked as a Scientific Review Officer at National Institute on Drug Abuse (NIDA). Dr. Hiremath received her Ph.D. in Microbiology and Immunology from the University of North Carolina at Chapel Hill.
- Julie Bronder Mason, Ph.D., joined NIMH as the Deputy Director of the Office of Science Policy, Planning, and Communications (OSPPC). Dr. Mason comes to NIMH with over 13 years of NIH experience. She most recently served as Associate Director in the Center for Cancer Training at the National Cancer Institute, where she led training program development and evaluation, workforce analyses, strategic planning, and communications. Dr. Mason earned her Ph.D. in Pharmacology and Toxicology from the Medical College of Virginia.
- Beverly Pringle, Ph.D., is the new Deputy Director for the Office of Research on Disparities and Global Mental Health (ORDGMH). Dr. Pringle will also continue to serve NIMH as Chief of Global Mental Health Research. She joined NIMH’s Division of Services and Intervention Research in 2009. In 2012, Dr. Pringle worked with the Centers for Disease Control and Prevention (CDC), providing technical assistance to staff of the Mozambique Ministry of Health and of the Mozambique CDC. Previously, Dr. Pringle served NIH as Chief of the Services Research Branch in the Division of Epidemiology, Services, and Prevention Research at NIDA. Dr. Pringle received her Ph.D. in Clinical Psychology from the University of Maryland, Baltimore County.
- The Division of Intramural Research Programs (IRP) notes changes in leadership and staff awards.
- Mortimer Mishkin, Ph.D., retired from the IRP faculty as of December 31, 2016, and returned in January 2017 as Scientist Emeritus in the Laboratory of Neuropsychology. Dr. Mishkin has been an IRP Principal Investigator since 1955. He headed the Section on Cognitive Neuroscience and the Laboratory of Neuropsychology, and served as Associate Director for Basic Research in the NIMH IRP (1994-1997). In his research, Dr. Mishkin used a variety of approaches to investigate the neurobiological mechanisms underlying learning and memory.
- Arash Afraz, M.D., Ph.D., joined the NIMH IRP as a tenure-track investigator and Chief of the Unit on Neurons, Circuits and Behavior. Dr. Afraz is interested in combining interventional physiology (microstimulation, drug microinjection, and optogenetics) with neural recordings to study the functional circuitry of the neural networks that drive object recognition. Using these techniques, he previously found that precisely timed electrical stimulation of face-selective neurons in inferior temporal cortex can bias behavioral choices in a face-detection task.
- Samer Hattar, Ph.D., joined the NIMH IRP as a Senior Investigator and Chief of the Section on Light and Biological Rhythms. Dr. Hattar comes to the NIMH from the Johns Hopkins University and School of Medicine, where he held joint Associate Professorships in the Departments of Biology and Neuroscience. His research helped define the retinal circuits and brain centers by which intrinsically photosensitive retinal ganglion cells (a type of neuron in the eye that signal the presence of light over the long term) drive a variety of critical functions, such as pupillary light reflex, circadian response to light and dark, vision, sleep, and mood.
- Karen F. Berman, M.D., Senior Investigator and Chief of the Clinical and Translational Neuroscience Branch, was elected to the National Academy of Medicine (NAM), which is one of the highest honors in the fields of health and medicine.
- Francis McMahon, M.D., Chief of the Human Genetics Branch, was named a recipient of the Brain & Behavior Research Foundation’s 2016 Colvin Prize for Outstanding Achievement in Mood Disorders Research.
- Miles Herkenham, Ph.D., Chief of the Section on Functional Neuroanatomy, received the 2017 Winter Conference on Brain Research Pioneer Award.
Director’s Highlights: NIMH Scientists and Science
Grantee Awards
NIMH is proud to recognize significant achievement and awards received by our current grantees:
- On January 9, 2017, President Obama announced the 2017 Presidential Early Career Awards for Scientists and Engineers (PECASE) , the highest honor bestowed by the United States Government on science and engineering professionals in the early stages of their independent research careers. Two of the awardees are NIMH grantees:
- Eric Morrow, M.D., Ph.D. (Brown University)
- Mary Kay Lobo, Ph.D. (University of Maryland School of Medicine)
- 2016 Troland Research Award, National Academy of Sciences
- David J. Freedman, Ph.D. (University of Chicago)
- Geoffrey F. Woodman, Ph.D. (Vanderbilt University)
- 2016 Brain & Behavior Research Foundation Awards, NIMH top scientists recognized
- Colvin Prize for Outstanding Achievement in Mood Disorders Research
- Thomas Schulze, M.D. (University of Munich)
- Pamela Sklar, M.D. (Icahn School of Medicine at Mount Sinai)
- Pardes Humanitarian Prize in Mental Health
- Vikram Patel, Ph.D. (Sangath)
- Charles F. Reynolds, III, M.D. (University of Pittsburgh)
- Goldman-Rakic Prize for Outstanding Achievement in Cognitive Neuroscience
- Earl K. Miller, Ph.D. (Massachusetts Institute of Technology)
- Ruane Prize for Outstanding Achievement in Child and Adolescent Psychiatric Research
- John L.R. Rubenstein, M.D., Ph.D. (University of California at San Francisco)
- Lieber Prize for Outstanding Achievement in Schizophrenia Research
- Michael F. Green, Ph.D. (University of California at Los Angeles)
- Stephen R. Marder, M.D. (University of California at Los Angeles)
- Maltz Prize for Innovative and Promising Schizophrenia Research
- William P. Horan, Ph.D. (University of California at Los Angeles)
- Amanda McCleery, Ph.D. (University of California at Los Angeles)
- Freedman Prize for Exceptional Basic Research
- Kay Tye, Ph.D. (Massachusetts Institute of Technology)
- Colvin Prize for Outstanding Achievement in Mood Disorders Research
- 2016 Society for Neuroscience (SfN) Young Investigator Award
- Kay Tye, Ph.D. (Massachusetts Institute of Technology)
- American Psychiatric Association (APA) Foundation’s 2016 Alexander Gralnick, M.D. Award
- John Kane, M.D. (Zucker Hillside Hospital)
- APA 2016–2017 Psychiatric Services Achievement Award – Silver Award
- OnTrackNY (Washington Heights Community Service)
- 2017 Winter Conference on Brain Research (WCBR) Pioneer Award
- Roger Nicoll, M.D. (University California San Francisco)
Notable NIMH Grants
The following is a selection of the Institute’s most recently funded projects that exemplify our efforts to accelerate mental health research and to advance the NIMH Strategic Plan for Research:
- Microglia, the brain’s resident immune cells, are essential for normal brain function, playing a role in synapse elimination, circuit formation, and injury response and repair. New evidence is beginning to define specific pathways by which microglial dysfunction may contribute to mental disorders. Currently, research on these pathways is limited by the fact that postmortem human microglia in culture rapidly lose their microglial identity. Frederick Bennett, M.D. (Stanford University), proposes to develop an innovative system that induces transplanted human blood cells to differentiate into microglia-like cells in the brains of transgenic mice. This novel system could be used to study human microglial biology in vivo, which would be broadly useful to researchers. This work aims to improve our understanding of the basic biology and development of microglia, and may offer a new method for studying their function in mental disorders.
- Following a traumatic event, it is common for people to report a range of symptoms, including hypervigilance, intrusive upsetting thoughts, flashbacks, and changes in sleep and mood. These often co-occur with chronic pain and substance use. Currently, there is no reliable way to predict who will recover without treatment and who will develop lasting problems after trauma. To address this, Samuel McLean M.D., M.P.H. (University of North Carolina), Ronald Kessler, Ph.D. (Harvard), Karestan Koenen, Ph.D. (Harvard), and Kerry Ressler M.D., Ph.D. (Emory), are leading a multisite longitudinal assessment of post-traumatic syndromes that will track 5,000 people after they have experienced a traumatic event. Over a year, researchers will collect biological data, use mobile technology to monitor participants’ activity, sleep, and mood, and administer in-depth exams that include functional brain imaging and psychological tests. Data from this study may provide a much deeper understanding of the mechanisms that give rise to post-traumatic disorders, as well as a clearer basis for predicting who will be affected and how best to target treatment.
- Recently, NIMH funded three new grants that support the Zero Suicide commitment to preventing suicide attempts and deaths among individuals receiving treatment within health care systems. Barbara Stanley, Ph.D. (Columbia University), plans to compare quality improvements in suicide prevention practice across 145 outpatient state licensed clinics that represent 85 New York state agencies, and include 1,490 clinical providers that reach over 80,000 adult clients. The second project – System of Safety (SOS)—builds off what was learned from the recently completed NIMH ED-SAFE study. ED-SAFE focused on adults who were at risk for suicide and went to an emergency department (ED) for care. With SOS, Edwin Boudreaux, Ph.D. , and Catarina Kiefe, M.D., Ph.D. (University of Massachusetts), will extend the ED-SAFE continuous quality improvement approach. They plan to examine the benefits of improved brief suicide-risk screening, providing outpatient suicide prevention discharge resources, and follow-up telephone counseling for the patient and a significant other. The third funded Zero Suicide study focuses on youth at risk for suicidal behavior within the Kaiser Permanente health system. Joan Asarnow, Ph.D. (University of California Los Angeles), and Greg Clarke Ph.D. (Kaiser Permanente), are leading a randomized trial of stepped care for suicide prevention in teens and young adults.
For more information on these and other grants selected for funding, please visit the NIH RePORTER website .
Current Funding Opportunities and Announcements
NIH electronically posts the NIH Guide , a listing of all NIH Funding Opportunity Announcements (FOAs) that includes requests for applications (RFAs), program announcements (PAs), and important notices for the scientific community. Below is a selection of recently issued FOAs in which NIMH participates. The Funding page on the NIMH website has links to listings of all NIMH FOAs and other resources.
You can subscribe to the NIMH Funding Opportunities ListServ to receive the latest information about RFAs and other research funding opportunities from NIMH, as well as administrative updates and changes to grant policies and procedures. You can also subscribe to a separate listserv to receive weekly e-mails from the NIH Guide .
NIMH-Administered Requests for Applications
- Using the NIMH Research Domain Criteria (RDoC) Approach to Understand Psychosis
- Release date: March 17, 2016; Standard due dates apply; Expiration date: (R01) March 6, 2017, (R21) March 17, 2017
- R01 announcement (PAR-16-136 )
- R21 announcement (PAR-16-135 )
- BRAIN Initiative Fellows: Ruth L. Kirschstein National Research Service Award (NRSA) Individual Postdoctoral Fellowship
- Release date: September 19, 2016; Application due date: March 15, 2017
- F32 announcement (RFA-MH-17-250 )
- Limited Competition: Continuation of the Center for Genomic Studies on Mental Disorders
- Release date: February 3, 2017; Application due date: April 7, 2017
- U24 announcement (RFA-MH-18-100 )
- Implementation Science for the Prevention and Treatment of Mental and/or Substance use Disorders in Low- and Middle-income Countries
- Release date: December 23, 2016; Application due date: April 7, 2017
- U01 announcement (RFA-MH-17-650 )
- BRAIN Initiative: Standards to Define Experiments Related to the BRAIN Initiative
- Release date: September 19, 2016; Application due date: October 11, 2017
- R24 announcement (RFA-MH-17-256 )
- BRAIN Initiative: Development and Validation of Novel Tools to Analyze Cell-Specific and Circuit-Specific Processes in the Brain
- Release date: August 10, 2016; Application due date: October 13, 2017
- R01 announcement (RFA-MH-17-220 )
- BRAIN Initiative: Foundations of Non-Invasive Functional Human Brain Imaging and Recording –Bridging Scales and Modalities
- Release date: August 15, 2016; Application due date: October 13, 2017
- R01 announcement (RFA-MH-17-235 )
- BRAIN Initiative: Non-Invasive Neuromodulation - Mechanisms and Dose/Response Relationships for Targeted CNS Effects
- Release date: August 23, 2016; Application due date: October 13, 2017
- R01 announcement (RFA-MH-17-245 )
- BRAIN Initiative: Non-Invasive Neuromodulation - New Tools and Techniques for Spatiotemporal Precision
- Release date: August 23, 2016; Application due date: October 13, 2017
- R01 announcement (RFA-MH-17-240 )
- BRAIN Initiative Cell Census Network (BICCN) - Specialized Center on Human and Non-Human Primate Brain Cell Atlases
- Release date: October 19, 2016; Application due date: October 13, 2017
- U01 announcement (RFA-MH-17-210 )
- BRAIN Initiative Cell Census Network (BICCN) - Specialized Center on Mouse Brain Cell Atlas
- Release date: October 19, 2016; Application due date: October 13, 2017
- U01 announcement (RFA-MH-17-230 )
- BRAIN Initiative: Data Archives for the BRAIN Initiative
- Release date: September 19, 2016; Application due date: October 19, 2017
- R24 announcement (RFA-MH-17-255 )
- BRAIN Initiative: Integration and Analysis of BRAIN Initiative Data
- Release date: September 21, 2016; Application due date: October 26, 2017
- R24 announcement (RFA-MH-17-257 )
- Early Stage Testing of Pharmacologic or Device-based Interventions for the Treatment of Mental Disorders
- Release date: December 13, 2016; Application due dates: February 15, 2017 - October 15, 2018
- R61/R33 announcement (RFA-MH-17-600 )
- R33 announcement (RFA-MH-17-602 )
- Development of Psychosocial Therapeutic and Preventive Interventions for Mental Disorders
- Release date: December 13, 2016; Application due dates: February 15, 2017 - October 15, 2018
- R61/R33 announcement (RFA-MH-17-604 )
- R33 announcement(RFA-MH-17-606 )
- Confirmatory Efficacy Clinical Trials of Non-Pharmacological Interventions for Mental Disorders
- Release date: December 13, 2016; Application due dates: February 15, 2017 - October 15, 2018
- R01 announcement (RFA-MH-17-614 )
- Pilot Effectiveness Trials for Treatment, Preventive and Services Interventions
- Release date: December 13, 2016; Application due dates: February 15, 2017 - October 15, 2018
- R34 announcement (RFA-MH-17-612 )
- Clinical Trials to Test the Effectiveness of Treatment, Preventive, and Services Interventions
- Release date: December 13, 2016; Application due dates: February 15, 2017 - October 15, 2018
- Collaborative R01 announcement (RFA-MH-17-610 )
- R01 announcement (RFA-MH-17-608 )
- Reducing the Duration of Untreated Psychosis in the United States
- Release date: May 17, 2016; Standard due dates apply; Expiration date: March 20, 2019
- R01 announcement (PAR-16-265 )
- R34 announcement (PAR-16-264 )
NIMH-Collaborative Requests for Applications
- BRAIN Initiative: Exploratory Targeted BRAIN Circuits Projects - eTargetedBCP
- Release date: December 2 2016; Application due date: March 8, 2017
- R21 announcement (RFA-NS-17-015 )
- BRAIN Initiative: Targeted BRAIN Circuits Projects - TargetedBCP
- Release date: December 2, 2016; Application due date: March 8, 2017
- R01 announcement (RFA-NS-17-014 )
- Revision Applications for U.S-South Africa Program for Collaborative Biomedical Research
- Release date: December 8, 2016; Application due date: March 28, 2017
- R01 announcement (RFA-AI-16-082 )
- NIH Blueprint Diversity Specialized Predoctoral to Postdoctoral Advancement in Neuroscience (D-SPAN) Award
- Release date: November 1, 2016; Application due date: April 8, 2017
- F99/K00 announcement (RFA-NS-17-009 )
- BRAIN Initiative: Research Career Enhancement Award for Investigators to Build Skills in a Cross-Disciplinary Area
- Release date: October 3, 2016; Application due date: April 14, 2017
- K18 announcement (RFA-DA-17-022 )
- NINDS Data Management and Coordinating Center (DMCC) for the Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) Collaborative Research Centers (CRCs)
- Release date: January 27, 2017; Application due date: May 2, 2017
- U24 announcement (RFA-NS-17-022 )
- NINDS Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) Collaborative Research Centers (CRCs)
- Release date: January 27, 2017; Application due date: May 2, 2017
- U54 announcement (RFA-NS-17-021 )
- BD2K Open Educational Resources for Skills Development in Biomedical Big Data Science
- Release date: May 18, 2016; Application due date: August 2, 2017
- R25 announcement (RFA-HG-16-016 )
- BRAIN Initiative: Team-Research BRAIN Circuit Programs - TeamBCP
- Release date: December 2, 2016; Application due dates: March 1, 2017; October 17, 2017
- U19 announcement (RFA-NS-17-018 )
- BRAIN Initiative: Clinical Studies to Advance Next-Generation Invasive Devices for Recording and Modulation in the Human Central Nervous System
- Release date: September 29, 2016; Application due date: October 18, 2017
- UH3 announcement (RFA-NS-17-006
- BRAIN Initiative: Next-Generation Invasive Devices for Recording and Modulation in the Human Central Nervous System
- Release date: September 29, 2016; Application due date: October 18, 2017
- U44 announcement (RFA-NS-17-007 )
- UG3/UH3 announcement (RFA-NS-17-005 )
- BRAIN Initiative: New Technologies and Novel Approaches for Large-Scale Recording and Modulation in the Nervous System
- Release date: September 29, 2016; Application due date: October 18, 2017
- U01 announcement (RFA-NS-17-003 )
- BRAIN Initiative: Optimization of Transformative Technologies for Large Scale Recording and Modulation in the Nervous System
- Release date: September 29, 2016; Application due date: October 18, 2017
- U01 announcement (RFA-NS-17-004 )
- BRAIN Initiative: SBIR Direct to Phase II Next-Generation Invasive Devices for Recording and Modulation in the Human Central Nervous System
- Release date: September 29, 2016; Application due date: October 18, 2017
- U44 announcement (RFA-NS-17-008 )
- Big Data to Knowledge (BD2K) Community-based Data and Metadata Standards Efforts
- Release date: August 16, 2016; Application due date: October 19, 2017
- R24 announcement (RFA-ES-16-010 )
- BD2K Research Education Curriculum Development: Data Science Overview for Biomedical Scientists
- Release date: August 10, 2016; Application due date: December 1, 2017
- R25 announcement (RFA-ES-16-011 )
- BD2K Support for Meetings of Data Science Related Organizations
- Release date: October 7, 2016; Application due date: November 30, 2018
- U13 announcement (RFA-CA-16-020 )
Future Research Directions
NIMH-Sponsored Meetings
- Leveraging Electronic Medical Records (EMR) for Psychiatric Genetic Research Workshop: The application of EMR-based genomics to mental illnesses offers substantial opportunities but also raises a number of challenges. On September 15, 2016, NIMH hosted a workshop to bring together leaders and experts in genomics, informatics, big data analytics, and psychiatric phenotyping to address these challenges. The workshop included presentations and discussion of key issues in leveraging EMR data for psychiatric genetic research, lessons learned from ongoing national and international efforts in EMR genomics, and descriptions of available and emerging resources comprising psychiatric phenotypes and genomic data. The workshop also addressed prospects for future collaborative research projects and opportunities.
- NIMH Alliance for Research Progress: NIMH convened the 23rd meeting of the Alliance for Research Progress on September 23, 2016. Alliance participants represent wide-ranging perspectives, including those of consumers, providers of mental health services, and family members. This annual gathering provides an opportunity for attendees to network with their colleagues in the advocacy community and to interact directly with the NIMH Director and other senior NIMH staff. The meeting presentations focused on the Emergency Department Screening for Teens at Risk for Suicide (ED-STARS), the NIH Precision Medicine Initiative Cohort Study (recently renamed the All of Us Research Program ), improving outcomes for youth with bipolar disorder, and suicide prevention after jail detention.
- NIMH Transcranial Electrical Stimulation (tES) Workshop: The NIMH Division of Translational Research sponsored a workshop titled Transcranial Electrical Stimulation (tES): Mechanisms, Technology, and Therapeutic Applications on September 29-30, 2016. The goal was to critically assess the use and potential of contemporary noninvasive neuromodulation techniques – particularly transcranial direct current stimulation (tDCS) and transcranial alternating current stimulation (tACS), which have been shown to affect physiology, brain function, and behavior. The workshop brought together experts in basic and human neuroscience, electrical technologies, and clinicians. Participants examined the physiological mechanisms underlying tDCS/tACS effects, technologies, and technical strategies for optimizing stimulation protocols. They also assessed the state of the science and mapped out future directions and the avenues for enhancing rigor and reproducibility in tDCS/tACS research.
- Research Domain Criteria (RDoC) Principal Investigators’ Meeting: On October 17-18, 2016, the NIMH RDoC Unit hosted a meeting to mark the fifth anniversary of the first RDoC funding opportunity announcement (FOA). The meeting brought together the principal investigators of grants funded under the first two RDoC R01 FOAs. Attendees came together to discuss their methodologies, collaborate, share their lessons learned, and discuss the future of RDoC with each other and with NIMH staff. Over 40 grants have been funded under RDoC FOAs since 2011.
- Webinar Series on the Science-to-Service Pipeline in Psychology and Psychiatry: On November 17, 2016, the NIMH RDoC Unit, the Delaware Project, and the Association for Behavioral and Cognitive Therapies (ABCT) launched a collaborative webinar series focused on the science-to-service pipeline in psychology and psychiatry. The first webinar in this series highlighted the role of fear and anxiety in disorders such as phobias and depression. Presenters with various perspectives discussed topics including basic laboratory research, treatment in clinics, and graduate school instruction for clinicians.
- NIMH Workshop: Using Stem Cell-Based Assays for Biological and Drug Discovery: On November 17-18, 2016, NIMH convened a workshop of scientists representing several ongoing funded research efforts involving human induced pluripotent stem cells (iPSCs) or similar reprogrammed cell assays. Participants represented the National Cooperative Reprogrammed Cell Research Groups (NCRCRG) , PsychENCODE , the Psychiatric Gene Networks initiative, the Brain Somatic Mosaicism initiative, and the NIH Common Fund Tissue Chip Consortium , along with a large number of investigator-initiated projects. Topics discussed included patient cell line collections versus genetically engineered isogenic cell lines in identifying genetically complex mechanisms underlying mental illnesses; measures needed to assess iPSC-derived cell-type fidelity and physiological relevance to their in vivo counterparts; and, needed improvements in cell assay technologies. Organizers are developing a summary of recommendations from this third NIMH iPSC grantee workshop, which will be made available to the scientific community.
- The NIMH IRP hosted three workshops and one symposium on the NIH Campus in Bethesda.
- Critical Brain Dynamics 2016: On October 17-19, 2016, NIMH hosted the fifth annual international workshop on criticality and the brain. The workshop focused on how modelling approaches complement experimental evidence and the impact of these insights towards clinical applications such as sleep disorders, epilepsy, and schizophrenia.
- A Celebration of 60 Years of Behavioral and Cognitive Neuroscience in the Laboratory of Neuropsychology: On October 20-21, 2016, NIMH hosted a symposium to celebrate the advances made over the last 60 years in the understanding of the neurobiology of memory, perception, and action. Speakers shared insights from studies using deficit-lesion methods, electrophysiology, imaging, and related techniques.
- Magnetoencephalography (MEG) North America Workshop: On November 1-2, 2016, the NIMH MEG Core Facility hosted a workshop to foster collaboration and synergy across MEG laboratories. Discussion focused on of MEG’s contribution to the field of neuroscience; the evolution of MEG acquisition and methods; development of infrastructure to share data and facilitate big science; and forming a MEG-North America consortium to address these topics.
Electronic Research Administration (eRA) Activities
Electronic Grant Application Submission News
- eRA is piloting a “virtual meeting” capability within Internet Assisted Review (IAR) so that review meetings may be held online, in lieu of in-person meetings. Additional details about IAR Virtual meeting are available for both reviewers and staff.
NIH-Wide Grant News
- NIH published a revised Grants Policy Statement . The October 2016 revisions update the October/November 2015 version and are applicable to all NIH grants and cooperative agreements beginning on or after October 1, 2016. The update incorporates new and modified requirements, clarifies certain policies, and implements changes in statutes, regulations, and policies that have been implemented through appropriate legal and/or policy processes since the previous version.
For more information on all of these updates, please see the NIH eRA News and Events page .
Questions? Contact the eRA help desk . Note that contacting this help desk is the only way to document problems with an electronic grant application submission. Evidence of this contact is the only way to be eligible for any special consideration by the Center for Scientific Review (CSR) Division of Receipt and Referral, should you run into a system problem with Grants.gov or with eRA that is beyond your control.
Research Training and Career Development
Here is the latest news about Research Training and Career Development at NIMH and NIH:
- The ability of NIH to help ensure that the nation remains a global leader in scientific discovery and innovation depends upon a pool of highly talented scientists from diverse backgrounds who will help to further NIH's mission (NOT-OD-15-053 ). The new NIH website, Diversity in Extramural Programs , provides a wealth of useful information in one location, organized under four themes: why workforce diversity matters; efforts to build participation; career pathways including funding options; and, reports and data. For example, T32 and R25 program directors can find strategies for strengthening their program’s recruitment plan, and individuals can explore career pathways and view videos of NIH-funded researchers.
- NIMH reissued three Research Education FOAs: Mentoring Networks for Mental Health Research Education (PAR-17-080 ); Research Education Programs for Psychiatry Residents (PAR-17-081 ); and, Short Courses for Mental Health-Related Research Education (PAR-17-082 ). Each FOA has a specific goal that falls under the over-arching goal of complementing other formal training programs within the mission of the NIMH. Please contact the NIMH Program staff listed in the FOA with questions. Applications for these FOAs are due annually on May 24.
- NIH reissued the FOA for the Jointly Sponsored National Research Service Award for Institutional Predoctoral T32 Programs in the Neurosciences (PAR-17-096 ). This funding opportunity supports broad and fundamental research training in the neurosciences during the first two years of graduate training, including a curriculum that provides a strong foundation in experimental design, statistical methodology, and quantitative reasoning. Please contact the NIMH Office of Research Training and Career Development with any questions. Applications are accepted annually on May 25.
Please refer to the NIMH webpage for research training and career development between issues of Inside NIMH to locate the latest news and resources for potential applicants and current awardees.
We are interested in feedback from the community; comments or suggestions related to NIMH’s support for research training and career development may be directed to NIMH_Training@mail.nih.gov. You may also contact NIMH Program Staff with questions or comments.
Director’s Messages
The Director’s Messages provide insights into the latest topics in mental health research:
- Computational Neuroscience: Deciphering the Complex Brain (February 7, 2017): Dr. Gordon explains how computational and theoretical neuroscience can advance research into mental disorders.
- Neural Circuits Research: How and Why (January 4, 2017): Dr. Gordon discusses the potential pay-offs of research on neural circuits and next steps for moving the science forward.
- The Push for Suicide Prevention (December 5, 2016): Dr. Gordon identifies suicide prevention as a priority research area and describes how ongoing efforts offer the potential to substantially reduce the suicide rate.
- Freshman Year (November 10, 2016): Dr. Gordon talks about his plans for his first year as NIMH Director, and his priorities for research.
NIMH Science News
The latest news and updates from NIMH-supported research:
- Revealed: LSD Docked in its Human Brain Target (February 1, 2017)
- Two NIMH Grantees Receive Prestigious Presidential Award (January 17, 2017)
- Sex Hormone–Sensitive Gene Complex Linked to Premenstrual Mood Disorder (January 3, 2017)
- Adding Better Mental Health Care to Primary Care (December 30, 2016)
- NIMH Training Grant Recipient Wins Research Prize (December 27, 2016)
- Molecular Tool Parses Social Fear Circuit Intertwined with Aggression Hub (November 30, 2016)
- Worldwide Study Seeks to Unlock the Brain’s Genetic Code (November 17, 2016)
- NIMH’s Francis McMahon, M.D., Awarded Prestigious Colvin Prize (November 1, 2016)
- RDoC Unit to Co-host Webinar Series (October 31, 2016)
- NIMH’s Karen F. Berman, M.D. elected to National Academy of Medicine (October 31, 2016)
- NIMH-Funded Study to Track the Effects of Trauma (October 17, 2016)
- NIH Nearly Doubles Investment in BRAIN Initiative Research (October 13, 2016)
- NIMH Releases Strategic Research Priorities Update (October 12, 2016)
- RDoC Unit to Host Virtual “Office Hours” (October 3, 2016)
- Powered-Up Probe ID’s Schizophrenia Genes That Stunt Brain Development (September 27, 2016)
- NIMH Funds 3 ‘Zero Suicide’ Grants (September 16, 2016)
Publicizing NIMH research is a communal responsibility. Please help us spread the word about the results of NIMH funding by acknowledging our support of your research, for example, in journal articles (citing your NIMH award by number when possible) and other communications. NIMH has two primary methods of getting the word out: press releases and science updates. All releases and updates are posted to the Science News section of the NIMH Web site. These are also distributed to the public through a mailing list .
Connect with NIMH
Inside NIMH is produced by the National Institute of Mental Health. For more information about the Institute, visit our website at https://www.nimh.nih.gov. For comments and suggestions about Inside NIMH, please contact the NIMH Webmaster. The material in this newsletter is not copyrighted, and we encourage its use or reprinting.
Sign up for the latest mental health news, research advances, upcoming events, publications, clinical trials, meeting summaries, and more. In addition to our email newsletters and RSS updates, please also visit NIMH on Twitter , Facebook , and YouTube , where we highlight Science Updates, Press Releases, and other timely matters.
