2018 Winter Inside NIMH

Welcome
Welcome to the latest edition of Inside NIMH! We publish Inside NIMH in conjunction with each meeting of the National Advisory Mental Health Council, which advises the Secretary of Health and Human Services (HHS), the Director of the National Institutes of Health (NIH), and the Director of NIMH on all policies and activities relating to the conduct and support of mental health research, research training, and other programs of the Institute. In addition, check out our website for regular updates on timely topics at NIMH. I hope you find this information interesting and helpful. Please let us know if you have questions or comments on this edition.
Sincerely,
Joshua A. Gordon, M.D., Ph.D.
Director, National Institute of Mental Health
If you wish to unsubscribe, subscribe, or change your email address, please contact the NIMH Webmaster or visit the Inside NIMH subscription page .
NIMH Director’s Updates
As we venture into 2018, it is a good time to reflect on our progress and our successes in 2017, my first full year at NIMH.
The Year in Review at NIMH
- Reflections and Future Directions: I spent my first year as Director of NIMH listening, learning, and finding my way. I have begun to share where we need to go as an Institute, in the context of a research portfolio that includes short-, medium-, and long-term investments, spanning disease-focused basic science and clinical research. I expressed my vision across an array of important topics including psychosocial interventions, autism spectrum disorder, NIMH Research Domain Criteria (RDoC), and critical next steps for suicide prevention. I also described my recent trip to the emergency room following a Thanksgiving football injury, noting the differences between being ill with a broken bone vs. having a mental illness. To date, I have been sharing this information through my Director’s Messages; I am happy to announce that I am now also sharing NIMH news and science more rapidly and in the moment through my recently launched Twitter account – @NIMHDirector .
- Strategic Research Priorities: To keep pace with the rapidly changing mental health research landscape, NIMH released our second annual update to the NIMH Strategic Research Priorities, which align with the NIMH Strategic Plan for Research. These priorities serve as guidance for NIMH grantees, potential grant applicants, and staff. The Strategic Research Priorities continue to emphasize rigor and reproducibility, and highlight the importance of data sharing and the use of common data elements. The updated priorities also encourage the use of computational approaches to further our understanding of the pathophysiology underlying mental illnesses.
- Redesigned Statistics Pages: NIMH revamped its statistics webpages, which include data on the prevalence and treatment of mental illnesses in the United States, as well as information about possible consequences of mental illnesses, such as suicide and disability. The redesigned pages feature better organization and navigation, compelling data visualization, downloadable graphics, an interactive user experience, easy sharing, and improved accessibility.
NIH Updates and Announcements
- Changes to Grant Application Processes: Effective January 25, 2018, all funding opportunity announcements require a new set of applications forms , FORMS-E. Researchers conducting human subjects studies must be careful to assess if the proposed research falls under the 2014 NIH definition of a clinical trial . Clinical trials applications will now be submitted under FOAs that allow for such trials, and the titles of all new FOAs now specify if clinical trial applications are accepted. NIMH accepts clinical trials applications through NIMH clinical trials FOAs, but only accepts “mechanistic” clinical trials under the NIH parent announcement for clinical trials .
- Interagency Autism Coordinating Committee (IACC) Updates: The IACC met on October 24, 2017, and announced the release of several publications. The 2016-2017 IACC Strategic Plan for Autism Spectrum Disorder (ASD) outlines 23 new objectives for ASD research and services activities, and includes a recommended ASD research budget. The 2014-2015 IACC Autism Spectrum Disorder Research Portfolio Analysis Report and corresponding update to the online Autism Research Database now feature data on federal and private autism research funding from 2008 through 2015.
- Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Updates:
- New BRAIN Initiative Awards: NIH announced funding for 110 new awards for the BRAIN Initiative . The new round of awards includes the BRAIN Initiative Cell Census Network (BICCN), aimed at providing researchers with comprehensive references of diverse brain cell types to generate knowledge necessary for understanding brain disorders. Additionally, new BRAIN Initiative neuroethics research awards aim to address neuroethical issues associated with human brain research.
- NIH BRAIN Initiative Director: NIH is actively seeking a Director for the NIH BRAIN Initiative. The BRAIN Initiative Director will lead scientific staff from the 10 NIH BRAIN Initiative Institutes and Centers, serve as spokesperson for the NIH BRAIN Initiative, and work with other federal and international agencies, as well as academic and commercial partners.
- All of Us Research Program:
- Outreach Efforts: All of Us announced a partnership with fourteen national community groups and health care provider associations to support outreach efforts for the program. These organizations will help raise awareness to engage one million or more volunteers across the country to build one of the largest, most diverse datasets of its kind for health research. This inaugural group will educate their communities about the benefits of participation in this landmark project to accelerate breakthroughs in precision medicine. The Program also announced a partnership with the National Library of Medicine to support outreach to communities through local libraries.
- Working Group Updates: The All of Us Genomics Working Group proposed a phased approach for genomic analyses, advising the program to evaluate both genotyping and whole-genome sequencing in a limited number of participants before attempting genomic analysis for all participants. The Program’s Advisory Panel established the Tribal Collaboration Working Group to provide input on the inclusion of American Indian and Alaska Native populations.
- Research Priorities: Through February 9, 2018, All of Us seeks input on its research priorities through “use cases” that describe a health problem or research question of interest. Submitted ideas will be considered at a Research Priorities Workshop in March 2018 aimed at identifying potential new program features to support research across a range of health topics.
- NIH leadership changes:
- In August 2017, Josie Briggs, M.D. , retired from her position as Director of the National Center for Complementary and Integrative Health (NCCIH). Dr. Briggs held several roles during her 20-year tenure at NIH, including inaugural lead of the Precision Medicine Initiative – now called All of Us. She is now Editor-in-Chief of the Journal of the American Society of Nephrology.
- In October 2017, Norman E. “Ned” Sharpless, M.D. , became the 15th Director of the National Cancer Institute (NCI). Dr. Sharpless came to NCI from the University of North Carolina School of Medicine, Chapel Hill, where he served as Director of the NCI-Designated Lineberger Comprehensive Cancer Center and as the Wellcome Distinguished Professor in Cancer Research.
- In November 2017, Roderic I. Pettigrew, Ph.D., M.D. , retired from his position as the founding Director of the National Institute of Biomedical Imaging and Bioengineering (NIBIB), after 15 years of service. Dr. Pettigrew will assume two senior leadership positions at Texas A&M University. The NIBIB Deputy Director, Jill Heemskerk, Ph.D., is serving as Acting Director.
Budget Overview
Fiscal Year (FY) 2017 Budget: NIMH awarded 571 new and competing research project grants (RPGs) in 2017, and achieved an overall success rate of approximately 21 percent (defined as number of RPG applications funded divided by the number of applications received; see Figures 1 and 2 below). NIMH awarded grants to 85 new investigators, and achieved a success rate of approximately 27 percent for Early Stage Investigators .
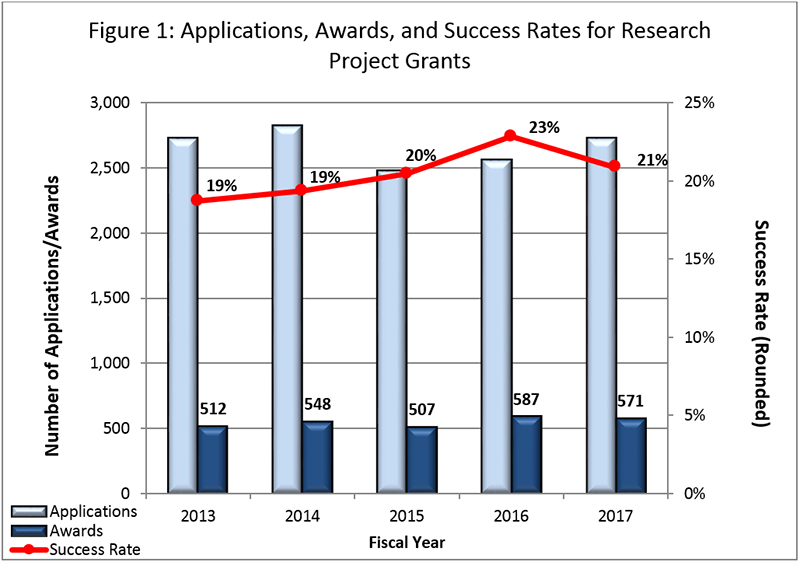
Figure 2 below shows the number of applications received and the number of awards made for R01 and equivalent competing grants in FY 2017. Data are presented using a new method developed by the NIH Office of Extramural Research showing success rates. The number of percentiled competing awards was 230 for $129M.
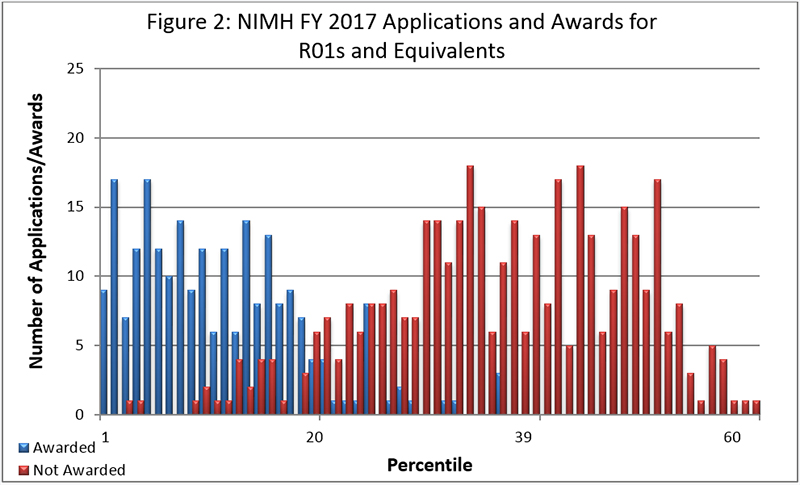
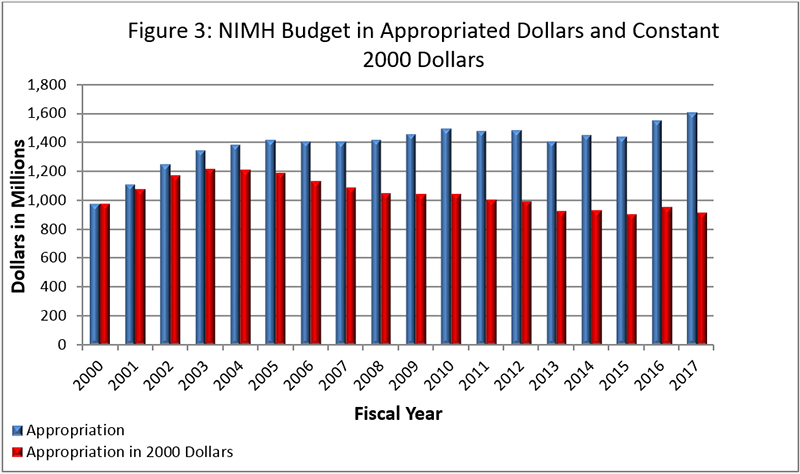
Figure 3 shows the NIMH budget in appropriated (current) versus constant (FY 2000) dollars. Constant dollars are “inflation adjusted” for variations in the purchasing power of the dollar over time. Dollar amounts are adjusted based on the Biomedical Research and Development Price Index (BRDPI). The annual change in BRDPI indicates how much the NIH budget must change to maintain purchasing power similar to FY 2000.
- Outlook for FY 2018: FY 2018 will begin under a continuing resolution (CR). As in the past, while operating under a CR, non-competing grants would be awarded at levels below committed amounts, likely at 90%. As in previous years when operating under a CR, the commitment level for NIMH grants would be determined after we receive a full-year appropriation for FY 2018.
NIMH Staff News and Awards
- Bruce Cuthbert, Ph.D., Director of the NIMH RDoC Unit, received the Society for Psychophysiological Research’s Award for Distinguished Contribution to Psychophysiology , and the American Psychological Association’s Meritorious Service Commendation. Dr. Cuthbert received both awards in recognition of his integrative work on cognitive and affective processes in anxiety and mood disorders, as well as his leadership in advancing novel approaches to study the classification of mental disorders through the NIMH RDoC initiative.
- Anna Ordonez, M.D., Deputy Director of the NIMH Office of Clinical Research, was elected to the rank of Distinguished Fellow by the American Academy of Child and Adolescent Psychiatry. Dr. Ordonez received this honor in recognition of her high level of training, skill, experience, and excellence in the field of child and adolescent psychiatry.
- Richard Coppola, Ph.D., retired from his position as the Director of the NIMH Intramural Research Program (IRP) Magnetoencephalography (MEG) Core, after more than four decades of service to NIMH. Dr. Coppola founded the MEG Core, and served as its Director since 2002. NIMH IRP scientist, Allison Nugent, Ph.D., is serving as Acting Director.
- We are sad to announce the passing of two outstanding colleagues:
- Dennis L. Murphy, M.D., passed away on September 23, 2017. Dr. Murphy was the former Chief of the Laboratory of Clinical Science in the NIMH Division of Intramural Research Programs. He was known for his research on the neurobiology of mood and anxiety disorders using molecular, neurochemical, and genetic techniques. A distinguished researcher and mentor at NIMH for over four decades, Dr. Murphy’s legacy includes over 100 students and fellows, many of whom went on to serve as chairs of psychiatry and basic science departments around the world.
- S. Charles ("Chuck") Schulz, M.D., passed away on October 1, 2017. Dr. Schulz was the former NIMH Schizophrenia Branch Chief and founding Co-director of the International Congress on Schizophrenia Research. During a career spanning four decades, he made formative contributions and sustained a passion for advancing schizophrenia research. Dr. Schulz was influential in facilitating the careers of many scientists early in their careers.
Director’s Highlights: NIMH Scientists and Science
Grantee Awards and Updates
NIMH is proud to recognize significant achievements and awards received by our current grantees:
- 2017 Sarnat International Prize in Mental Health, National Academy of Medicine
- Joseph T. Coyle, M.D. (Harvard Medical School)
- Matthew State, M.D., Ph.D. (University of California, San Francisco)
- 2017 Joel Elkes Research Award, American College of Neuropsychopharmacology
- Carrie Bearden, Ph.D. (University of California, Los Angeles)
- Diego Pizzagalli, Ph.D. (Harvard Medical School)
- 2017 Gruber International Research Award, Society for Neuroscience
- Laura Lewis, Ph.D. (Harvard University)
- Laura Lewis, Ph.D. (Harvard University)
- 2017 Maltz Prize for Outstanding Achievement in Schizophrenia Research, Brain and Behavior Research Foundation
- Deanna Kelly, Pharm.D., BCPP (University of Maryland School of Medicine)
NIMH is sad to announce the passing of remarkable colleagues:
- Pamela Sklar, M.D., Ph.D., passed away on November 20, 2017. Dr. Sklar was Chair of the Department of Genetics and Genomic Sciences at the Icahn School of Medicine at Mount Sinai. A long-time NIMH grantee, she was a clinician scientist dedicated to delineating genetic risk factors and molecular processes underlying psychiatric disorders. Dr. Sklar was a seminal figure in psychiatric genetics, and received the 2017 International Society of Psychiatric Genetics’ Ming Tsuang Lifetime Achievement Award.
- Ben Barres, M.D., Ph.D., passed away on December 27, 2017. Dr. Barres was Chair of the Department of Neurobiology at Stanford University School of Medicine. An NIMH grantee, Dr. Barres was acclaimed for his groundbreaking discoveries of the crucial roles played by glial cells, which revolutionized the field of neuroscience. He is also remembered as a tireless advocate for diversity and equity in the scientific workforce.
Notable NIMH Grants
The following is a selection of the Institute’s most recently funded projects that exemplify our efforts to accelerate research on mental illnesses, and to advance the NIMH Strategic Plan for Research.
- The Zero Suicide campaign seeks to improve health care systems’ ability to identify who is at risk for suicide, and to implement effective treatments and services for at-risk individuals; the aspirational goal is to reach zero suicides in health care settings. Robert Aseltine, Ph.D. (University of Connecticut) aims to analyze medical claims, electronic health records, and vital statistics registries to identify patients at risk for suicidal behavior across multiple health systems. Dr. Aseltine will provide data to clinicians on patient risk, and will seek their input on acceptance and utility of this approach for improving patient care. Brian Ahmedani, Ph.D., LMSW (Henry Ford Health System), and Greg Simon, M.D., M.P.H. (Kaiser Permanente Washington Health Research Institute) are planning a comprehensive evaluation of the Zero Suicide campaign across six health care systems that collectively serve over nine million people.
- NIMH recently funded projects under the Autism Centers of Excellence (ACE) Program. Katarzyna Chawarska, Ph.D. (Yale University) will lead a team that aims to investigate how brain connections in early development, including the prenatal and early postnatal periods, function as early indicators of ASD. Ami Klin, Ph.D. (Emory University) and colleagues will examine social interactions in very early infancy to detect the earliest signs of ASD, and will identify infant characteristics that predict future treatment response and outcomes. Kevin Pelphrey, Ph.D. (George Washington University) will lead group of researchers seeking to understand how ASD differs among girls and boys, particularly during the transition from adolescence into adulthood. Diana Robins, Ph.D. (Drexel University) and colleagues will evaluate the extent to which early developmental screening lowers the average age of ASD diagnosis and leads to earlier interventions and improved outcomes.
- ASD is more common among males than females, and ASD among males tends to be more severe and more often associated with comorbidities. Yet, reasons for these sex differences remain elusive. Lauren Weiss, Ph.D. (University of California, San Francisco) seeks to understand the genetic basis of male sex as a major risk factor for ASD prevalence, severity, and comorbidities. Dr. Weiss will assess genome-wide mutations and specific loci for sex differences, and will apply new information to ASD data. This project may result in novel insights into complex genetic mechanisms contributing to ASD with implications for treatment.
- Combination anti-retroviral therapy (ART) has significantly altered the trajectory of the AIDS epidemic. The next generation of ART therapies aim to achieve viral eradication and disease elimination. HIV cure is high priority area for NIH, and eradicating the CNS viral reservoir is a key step. Howard Gendelman, M.D. (University of Nebraska Medical Center) and colleagues plan to employ sequential use of nanoformulated ART (nanoART) and CRISPR/Cas9 – an advanced gene editing technique – in a mouse model that mimics human HIV infection and progression with a focus on the CNS. This project has potential to reach its aspirational goal to achieve a cure for HIV.
For more information on these and other grants selected for funding, visit the NIH RePORTER website .
Current Funding Opportunities and Announcements
NIH electronically posts the NIH Guide , a listing of all NIH funding opportunity announcements (FOAs) that includes requests for applications (RFAs), program announcements (PAs), and important notices for the scientific community. Below is a selection of recently issued FOAs in which NIMH participates. The Funding page on the NIMH website has links to listings of all NIMH FOAs and other resources.
You can subscribe to the NIMH Funding Opportunities ListServ to receive the latest information about NIMH funding opportunities, as well as administrative updates and changes to grant policies and procedures. You can also subscribe to a separate listserv to receive weekly e-mails from the NIH Guide .
Please refer to a specific FOA for submission instructions including applications due dates, award and eligibility information, agency contacts, and additional information.
NIMH-Administered Requests for Applications
- Clinical Trials to Test the Effectiveness of Treatment, Preventive, and Services Interventions (Clinical Trial Required)
- Release date: November 14, 2017; Application due dates: February 14, 2018 - October 15, 2018
- Collaborative R01 announcement (RFA-MH-18-700 )
- R01 announcement (RFA-MH-18-701 )
- Confirmatory Efficacy Clinical Trials of Non-Pharmacological Interventions for Mental Disorders (Clinical Trial Required)
- Release date: November 14, 2017; Application due dates: February 14, 2018 - October 15, 2018
- R01 announcement (RFA-MH-18-707 )
- Development of Psychosocial Therapeutic and Preventive Interventions for Mental Disorders (Clinical Trial Required)
- Release date: November 14, 2017; Application due dates: February 14, 2018 - October 15, 2018
- R61/R33 announcement (RFA-MH-18-704 )
- R33 announcement (RFA-MH-18-705 )
- Early Stage Testing of Pharmacologic or Device-based Interventions for the Treatment of Mental Disorders (Clinical Trial Required)
- Release date: November 14, 2017; Application due dates: February 14, 2018 - October 15, 2018
- R61/R33 announcement (RFA-MH-18-702 )
- R33 announcement (RFA-MH-18-703 )
- Pilot Effectiveness Trials for Treatment, Preventive and Services Interventions (Clinical Trial Required)
- Release date: November 14, 2017; Application due dates: February 14, 2018 - October 15, 2018
- R34 announcement (RFA-MH-18-706 )
- BRAIN Initiative: Tools to Facilitate High-Throughput Microconnectivity Analysis
- Release date: August 30, 2017; Application due date: November 13, 2018
- R01 announcement (RFA-MH-18-505 )
- Reducing the Duration of Untreated Psychosis in the United States (Clinical Trial Required)
- Release date: November 21, 2017; Standard due dates apply; Expiration date: March 20, 2019
- R01 announcement (PAR-18-233 )
- R34 announcement (PAR-18-232 )
- From Genomic Association to Causation: A Convergent Neuroscience Approach for Integrating Levels of Analysis to Delineate Brain Function in Neuropsychiatry
- Release date: April 11, 2017; Standard due dates apply; Expiration date: May 8, 2019
- Collaborative R01 announcement (PAR-17-252 )
- R01 announcement (PAR-17-253 )
- NIMH Biobehavioral Research Awards for Innovative New Scientists
- Release date: February 17, 2017; Application due dates: June 20, 2018 - June 20, 2019
- R01 announcement (RFA-MH-18-200 )
- BRAIN Initiative Fellows: Ruth L. Kirschstein National Research Service Award Individual Postdoctoral Fellowship
- Release date: November 7, 2017; Application due dates: March 15, 2018 - April 7, 2020
- F32 announcement (RFA-MH-18-510 )
- Innovative Mental Health Services Research Not Involving Clinical Trials
- Release date: April 28, 2017; Standard due dates apply; Expiration date: September 8, 2020
- R01 announcement (PAR-17-264 )
- First in Human and Early Stage Clinical Trials of Novel Investigational Drugs or Devices for Psychiatric Disorders (Clinical Trial Required)
- Release date: December 6, 2017; Standard due dates apply; Expiration Date: January 8, 2021
- U01 announcement (PAR-18-427 )
NIMH-Collaborative Requests for Applications
- BRAIN Initiative: Research Resource Grants for Technology Integration and Dissemination (Clinical Trial Not Allowed)
- Release date: September 20, 2017; Application due date: February 9, 2018
- U24 announcement (RFA-NS-18-005 )
- Next Generation Multipurpose Prevention Technologies (Clinical Trial Optional)
- Release date: September 12, 2017; Application due date: March 19, 2018
- R61/R33 announcement (RFA-AI-17-028 )
- Limited Competition: Clinical Research Sites for the MACS/WIHS Combined Cohort Study (MACS/WIHS-CCS) (Clinical Trial Not Allowed)
- Release date: December 22, 2017; Application due date: May 2, 2018
- U01 announcement (RFA-HL-19-008 )
- Limited Competition: Data Analysis and Coordination Center for the MACS/WIHS Combined Cohort Study (MACS/WIHS-CCS) (Clinical Trial Not Allowed)
- Release date: December 22, 2017; Application due date: May 2, 2018
- U01 announcement (RFA-HL-19-007 )
- BRAIN Initiative: Tools to Target, Identify and Characterize Non-Neuronal Cells in the Brain (Clinical Trial Not Allowed)
- Release date: September 22, 2017; Application due date: October 4, 2018
- R01 announcement (RFA-DA-18-018 )
- BRAIN Initiative: Development of Next Generation Human Brain Imaging Tools and Technologies (Clinical Trial Not Allowed)
- Release date: October 4, 2017; Application due date: December 11, 2018
- U01 announcement (RFA-EB-17-004 )
- BRAIN Initiative: Proof of Concept Development of Early Stage Next Generation Human Brain Imaging (Clinical Trial Not Allowed)
- Release date: October 4, 2017; Application due date: December 11, 2018
- R01 announcement (RFA-EB-17-003 )
- BRAIN Initiative: Targeted BRAIN Circuits Planning Projects – TargetedBCPP (Clinical Trial Not Allowed)
- Release date: December 14, 2017; Application due date: July 15, 2019
- R34 announcement (RFA-NS-18-014 )
- BRAIN Initiative: Theories, Models and Methods for Analysis of Complex Data from the Brain (Clinical Trial Not Allowed)
- Release date: October 12, 2017; Application due date: October 17, 2019
- R01 announcement (RFA-EB-17-005 )
- BRAIN Initiative: Biology and Biophysics of Neural Stimulation (Clinical Trial Optional)
- Release date: December 14, 2017; Application due date: October 6, 2020
- R01 announcement (RFA-NS-18-018 )
- BRAIN Initiative: Clinical Studies to Advance Next-Generation Invasive Devices for Recording and Modulation in the Human Central Nervous System (Clinical Trial Required)
- Release date: December 21, 2017; Application due date: October 21, 2020
- UH3 announcement (RFA-NS-18-023 )
- BRAIN Initiative: Next-Generation Invasive Devices for Recording and Modulation in the Human Central Nervous System (Clinical Trial Required)
- Release date: December 21, 2017; Application due date: October 21, 2020
- UG3/UH3 announcement (RFA-NS-18-021 )
- U44 announcement (RFA-NS-18-022 )
Future Research Directions
Concept Clearances for Potential New Research Initiatives
This listing of potential future initiatives is meant to provide the earliest possible alert to the field of our research interests and of potential upcoming announcements to solicit that research. While NIMH plans to proceed with these initiatives, their publication and timing are not certain and depend on sufficient funding. The titles and brief descriptions are consistent with the information available at the time of concept clearance. The resultant FOAs may differ from the concepts in the final wording of their titles or other aspects. To send questions about a specific concept, follow the “Submit Comments” link at the bottom of the description.
- Computational Models for Validating Dimensional Approaches to Psychopathology
- Development and Validation of Technologies for Rapid Isolation and Characterization of Extracellular Vesicles of Central Nervous System Origin
For more information, please see recent NAMHC-approved concepts, recent public venue-approved concepts, and past NAMHC meetings, which also contains links to meeting agendas, minutes, and Inside NIMH (Director’s Reports).
NIMH-Sponsored Meetings
- Multimodal Brain Stimulation Speaker Series: From May through November 2017, NIMH hosted a speaker series to bring together leaders in the field conducting research using functional imaging techniques and non-invasive brain stimulation such as transcranial magnetic stimulation (TMS) and transcranial direct current stimulation. Topics included the use of functional imaging to target deep brain structures with TMS, and multimodal brain stimulation as a biomarker to predict the TMS response. Archived talks are available online .
- Blood-Brain Barrier (BBB) and HIV Reservoirs in the CNS – Formation and Eradication: On September 13-15, 2017, NIMH hosted a meeting in conjunction with the International Symposium on Signal Transduction at the Blood-Brain Barriers. The session focused on research related to the role of the BBB in regulating HIV reservoirs in the CNS, and approaches for eradicating HIV by targeting the barrier. This session also provided opportunities for HIV CNS researchers to interact with BBB researchers from a variety of fields.
- Sleep and Neurodevelopment Workshop: Electrophysiologic Sleep Phenotyping (ESP): On September 18, 2017, NIMH convened a workshop on sleep and neurodevelopment. Participants included experts in pulmonary medicine, sleep medicine, neurology, child psychiatry, developmental psychology, psychiatric genetics, translational science, and bioengineering. Discussions addressed efforts to enhance the pace of discovery in this nascent field, including ESP as a mainstay of clinical assessment of children at risk for neurodevelopmental disorders.
- Autism in Girls and Women: Research demonstrates that ASD presents differently in males and females. Researchers seek to understand the biological differences between ASD in males and females, and to reevaluate the effectiveness of diagnostic tools and treatments for females with ASD. On September 19, 2017, NIMH held a panel discussion on ASD in girls and women. Experts spoke about biological aspects of sex differences in ASD, observable differences in phenotype between boys and girls with ASD, as well as personal and community experiences related to ASD in girls and women.
- NIMH Rare Genetic Diseases Workshop: On September 19, 2017, NIMH hosted a workshop on challenges and opportunities for exploring rare genetic syndromes as models to delineate the genetic risk architecture of mental illnesses and to translate findings into meaningful prevention and treatments. Experts discussed the state of genetic analysis, phenotyping, and mechanistic characterization. NIH Institute Directors presented current NIH efforts in rare genetic disease research, and approaches to advance the field through collaboration.
- NIMH Alliance for Research Progress Meeting: On September 23, 2017, NIMH convened the twenty-fourth meeting of the Alliance for Research Progress. Alliance participants included representatives from patient and family advocacy organizations, and this annual meeting provided a forum for attendees to network with colleagues in the advocacy community and to interact directly with the NIMH Director and other senior NIMH staff. Presenters discussed the opioid crisis , treatments for obsessive-compulsive disorder and depression, new family-centered approaches to improve child mental health services, and updates from NIMH.
- Transcranial Electrical Stimulation (tES) Mechanisms, Technology and Therapeutic Applications: On September 29-30, 2017, NIMH sponsored a workshop to discuss contemporary forms of low current transcranial electrical stimulation (tES), particularly transcranial direct current stimulation (tDCS), and transcranial alternating current stimulation (tACS). Experts met to discuss physiological mechanisms, protocol and technology optimization, therapeutic applications and trial designs, and future directions. A report from this workshop will be published.
- Mental Health: Youth in Transition: On October 2, 2017, NIMH partnered with the Embassy of Canada and the Canadian Institutes of Health Research to host a panel discussion on mental health challenges faced by youth during the transition to adulthood. Experts examined research-based evidence to improve youth mental health through reliable access to services, integration of care, and stronger suicide prevention efforts. Participants identified feasible approaches to enhance healthy transitions from youth to adulthood.
- NeuroHIV in the ART Era: On October 23-24, 2017, NIMH partnered with other NIH institutes to convene a meeting on neuropsychiatric effects of HIV and its medications. Experts discussed a variety of topics including: HIV-induced neuropsychiatric symptoms and clinical presentation; influence of co-morbidities on development and diagnosis of HIV CNS disease; impact of HIV-induced inflammation and neuroimmune mediators on the CNS; and, impact of HIV CNS reservoirs on both disease development and efforts aimed at a cure.
- Approach to Consortium Studies of CNS HIV Escape: On October 11-14, 2017, NIMH hosted a session of the seventh International Meeting on HIV Infection in the CNS. Presenters discussed laboratory and clinical definitions of cerebrospinal fluid (CSF) escape; practical issues for consortium studies; and, clinical and laboratory implications for integrating CSF escape into HIV persistence and eradication studies. A consensus definition of CSF escape was developed and will be published.
- NIH-Magnetoencephalography (MEG) North America Workshop: On November 8-9, 2017, NIMH hosted a workshop to foster collaboration and synergy among NIMH intramural and extramural-supported MEG laboratories, as well as clinical MEG sites. Participants discussed reproducibility and reliability, best practices, data sharing, and industry-academic partnerships. Resulting action items include enhanced collaboration and use of existing data repositories.
- Explainable Artificial Intelligence (XAI) Solutions Applied to Neural and Behavioral Data: On November 10, 2017, NIMH convened a workshop to discuss XAI solutions to guide basic and clinical interventions, and to inform understanding of complex brain-behavioral data in the context of neuro-behavioral modulation. Attendees explored a variety of topics including: application of XAI solutions to neuroscience and psychiatric datasets; efforts to draw XAI scholars to the fields of neuroscience and psychiatry; and, strategies to ensure open cross-talk among investigators in this nascent field.
- Biobehavioral Research Awards for Innovative New Scientists (BRAINS) Awards Ceremony: On November 12, 2017, NIMH held a ceremony at the Society for Neuroscience conference to honor the 2016-2017 BRAINS awardees. The NIMH Director opened the ceremony and emphasized the importance of the BRAINS program, which supports outstanding new scientists with innovative research focused on the recovery, prevention, and cure of mental illnesses. Awardees presented their research to NIMH leadership, program staff, and previous BRAINS awardees.
Electronic Research Administration (eRA) Activities
NIH-Wide Grant News
- New NIH "FORMS-E" Grant Application Instructions Available for Due Dates On or After January 25, 2018: Effective January 25, 2018, all grant applications must be submitted using the new FORMS-E, which consolidates human subjects and inclusion enrollment report information, and expands fields for clinical trial data (NOT-OD-17-119 ).
- Publication of the Revised NIH Grants Policy Statement (Rev. October 2017) for FY 2018: Published on October 1, 2017, the new NIH grants policy statement includes standard terms and conditions of awards, and extensive guidance for researchers pursuing grants (NOT-OD-18-005 ).
- NIH will Make the Project Outcomes Section of all Interim and Final Research Performance Progress Reports Submitted Available via the NIH RePORTER: Effective October 1, 2017, NIH grantee project outcomes will be published in NIH RePORTER to provide the public with concise summaries of project outcomes (NOT-OD-18-103 ).
For more information on all of these updates, please see the NIH eRA News and Events page .
Questions? Contact the eRA Service desk . Note that contacting this help desk is the only way to document problems with an electronic grant application submission. Evidence of this contact is the only way to be eligible for any special consideration by the Center for Scientific Review (CSR) Division of Receipt and Referral, should you run into a system problem with Grants.gov or with eRA that is beyond your control.
Director’s Messages
NIMH’s Director’s Messages provide insights into the latest topics in mental health research:
- On Being Ill (December 14, 2017): After an emergency room visit on Thanksgiving, Dr. Gordon reflects on treatments for physical injury compared with those for psychiatric illnesses.
- Sophomore Year (October 30, 2017): Dr. Gordon reflects on his first year at NIMH and outlines priorities for going forward.
- Suicide Prevention: Next Steps (September 15, 2017): Dr. Gordon outlines recent research results and future directions.
NIMH Science News
The latest news and updates from NIMH-supported research:
- Memory Gene Goes Viral (January 16, 2018)
- Intramural Researchers Develop Suicide Risk Screening Toolkit for Medical Settings (January 12, 2018)
- Different Approaches to Understanding and Classifying Mental Disorders (December 27, 2017)
- NIH Announces Awards for BRAIN Initiative Neuroethics Research (November 6, 2017)
- Brain’s Alertness Circuitry Conserved Through Evolution (November 2, 2017)
- NIH BRAIN Initiative Builds on Early Advances (October 23, 2017)
- NIH BRAIN Initiative Launches Cell Census (October 23, 2017)
- NIH Completes Atlas of Human DNA Differences that Influence Gene Expression (October 12, 2017)
- Life-Saving Post-ER Suicide Prevention Strategies are Cost Effective (September 15, 2017)
Publicizing NIMH research is a communal responsibility. Please help us spread the word about the results of NIMH funding by acknowledging our support of your research, for example, in journal articles (citing your NIMH award by number when possible) and other communications. NIMH has two primary methods of getting the word out: press releases and science updates. All releases and updates are posted to the Science News section of the NIMH Web site. These are also distributed to the public through a mailing list .
Connect with NIMH
Sign up for the latest mental health news, research advances, upcoming events, publications, clinical trials, meeting summaries, and more. In addition to our email newsletters and RSS updates, please also visit NIMH on Twitter , Facebook , and YouTube , where we highlight Science Updates, Press Releases, and other timely matters.
Inside NIMH is produced by the National Institute of Mental Health. For more information about the Institute, visit our website at https://www.nimh.nih.gov. For comments and suggestions about Inside NIMH, please contact the NIMH Webmaster. The material in this newsletter is not copyrighted, and we encourage its use or reprinting.
